| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cholinesterase |
|---|
| Ligand | BDBM50449214 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1298067 (CHEMBL3129498) |
|---|
| IC50 | 290±n/a nM |
|---|
| Citation |  Bautista-Aguilera, OM; Esteban, G; Bolea, I; Nikolic, K; Agbaba, D; Moraleda, I; Iriepa, I; Samadi, A; Soriano, E; Unzeta, M; Marco-Contelles, J Design, synthesis, pharmacological evaluation, QSAR analysis, molecular modeling and ADMET of novel donepezil-indolyl hybrids as multipotent cholinesterase/monoamine oxidase inhibitors for the potential treatment of Alzheimer's disease. Eur J Med Chem75:82-95 (2014) [PubMed] Article Bautista-Aguilera, OM; Esteban, G; Bolea, I; Nikolic, K; Agbaba, D; Moraleda, I; Iriepa, I; Samadi, A; Soriano, E; Unzeta, M; Marco-Contelles, J Design, synthesis, pharmacological evaluation, QSAR analysis, molecular modeling and ADMET of novel donepezil-indolyl hybrids as multipotent cholinesterase/monoamine oxidase inhibitors for the potential treatment of Alzheimer's disease. Eur J Med Chem75:82-95 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cholinesterase |
|---|
| Name: | Cholinesterase |
|---|
| Synonyms: | BCHE | Butyrylcholinesterase (BuChE) | CHLE_HORSE | Cholinesterase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 65643.35 |
|---|
| Organism: | Equus caballus (Horse) |
|---|
| Description: | P81908 |
|---|
| Residue: | 574 |
|---|
| Sequence: | EEDIIITTKNGKVRGMNLPVLGGTVTAFLGIPYAQPPLGRLRFKKPQSLTKWSNIWNATK
YANSCYQNTDQSFPGFLGSEMWNPNTELSEDCLYLNVWIPAPKPKNATVMIWIYGGGFQT
GTSSLPVYDGKFLARVERVIVVSMNYRVGALGFLALSENPEAPGNMGLFDQQLALQWVQK
NIAAFGGNPRSVTLFGESAGAASVSLHLLSPRSQPLFTRAILQSGSSNAPWAVTSLYEAR
NRTLTLAKRMGCSRDNETEMIKCLRDKDPQEILLNEVFVVPYDTLLSVNFGPTVDGDFLT
DMPDTLLQLGQFKRTQILVGVNKDEGTAFLVYGAPGFSKDNNSIITRKEFQEGLKIFFPR
VSEFGRESILFHYMDWLDDQRAENYREALDDVVGDYNIICPALEFTRKFSELGNDAFFYY
FEHRSTKLPWPEWMGVMHGYEIEFVFGLPLERRVNYTRAEEILSRSIMKRWANFAKYGNP
NGTQNNSTRWPVFKSTEQKYLTLNTESPKVYTKLRAQQCRFWTLFFPKVLELTGNIDEAE
REWKAGFHRWNNYMMDWKNQFNDYTSKKESCSDF
|
|
|
|---|
| BDBM50449214 |
|---|
| n/a |
|---|
| Name | BDBM50449214 |
|---|
| Synonyms: | CHEMBL3127977 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H39N3O |
|---|
| Mol. Mass. | 481.6716 |
|---|
| SMILES | Cn1c(CNCc2ccccc2)cc2cc(OCCCC3CCN(Cc4ccccc4)CC3)ccc12 |
|---|
| Structure | 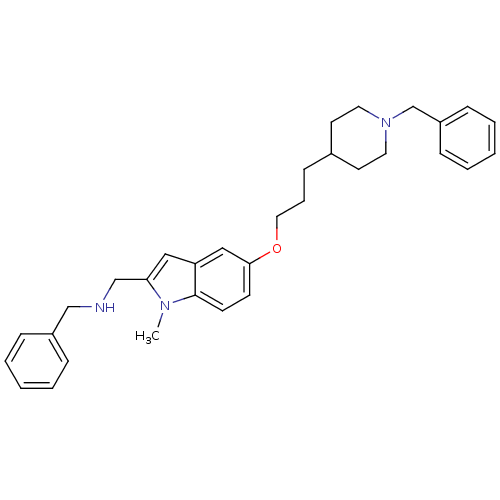
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bautista-Aguilera, OM; Esteban, G; Bolea, I; Nikolic, K; Agbaba, D; Moraleda, I; Iriepa, I; Samadi, A; Soriano, E; Unzeta, M; Marco-Contelles, J Design, synthesis, pharmacological evaluation, QSAR analysis, molecular modeling and ADMET of novel donepezil-indolyl hybrids as multipotent cholinesterase/monoamine oxidase inhibitors for the potential treatment of Alzheimer's disease. Eur J Med Chem75:82-95 (2014) [PubMed] Article
Bautista-Aguilera, OM; Esteban, G; Bolea, I; Nikolic, K; Agbaba, D; Moraleda, I; Iriepa, I; Samadi, A; Soriano, E; Unzeta, M; Marco-Contelles, J Design, synthesis, pharmacological evaluation, QSAR analysis, molecular modeling and ADMET of novel donepezil-indolyl hybrids as multipotent cholinesterase/monoamine oxidase inhibitors for the potential treatment of Alzheimer's disease. Eur J Med Chem75:82-95 (2014) [PubMed] Article