| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Amine oxidase [flavin-containing] B |
|---|
| Ligand | BDBM50087800 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1453699 (CHEMBL3364101) |
|---|
| IC50 | 1250±n/a nM |
|---|
| Citation |  Tzvetkov, NT; Hinz, S; K�ppers, P; Gastreich, M; M�ller, CE Indazole- and indole-5-carboxamides: selective and reversible monoamine oxidase B inhibitors with subnanomolar potency. J Med Chem57:6679-703 (2014) [PubMed] Article Tzvetkov, NT; Hinz, S; K�ppers, P; Gastreich, M; M�ller, CE Indazole- and indole-5-carboxamides: selective and reversible monoamine oxidase B inhibitors with subnanomolar potency. J Med Chem57:6679-703 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Amine oxidase [flavin-containing] B |
|---|
| Name: | Amine oxidase [flavin-containing] B |
|---|
| Synonyms: | AOFB_RAT | Amine oxidase (flavin-containing) B | Amine oxidase [flavin-containing] B | Maob | Monoamine Oxidase Type B (MAO-B) | Monoamine oxidase | Monoamine oxidase B (MAO-B) | Monoamine oxidase B (rMAO-B) | Monoamine oxidase type B (MAOB) | Monoamine oxidase-B (MAO-B) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58469.65 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | P19643 |
|---|
| Residue: | 520 |
|---|
| Sequence: | MSNKCDVIVVGGGISGMAAAKLLHDCGLSVVVLEARDRVGGRTYTIRNKNVKYVDLGGSY
VGPTQNRILRLAKELGLETYKVNEVERLIHFVKGKSYAFRGPFPPVWNPITYLDYNNLWR
TMDEMGQEIPSDAPWKAPLAEEWDYMTMKELLDKICWTNSTKQIATLFVNLCVTAETHEV
SALWFLWYVKQCGGTTRIISTTNGGQERKFIGGSGQVSERIKDILGDRVKLERPVIHIDQ
TGENVVVKTLNHEIYEAKYVISAIPPVLGMKIHHSPPLPILRNQLITRVPLGSVIKCMVY
YKEPFWRKKDFCGTMVIEGEEAPIAYTLDDTKPDGSCAAIMGFILAHKARKLVRLTKEER
LRKLCELYAKVLNSQEALQPVHYEEKNWCEEQYSGGCYTAYFPPGILTQYGRVLRQPVGK
IFFAGTETASHWSGYMEGAVEAGERAAREILHAIGKIPEDEIWQPEPESVDVPARPITNT
FLERHLPSVPGLLKLLGLTTILSATALGFLAHKKGLFVRF
|
|
|
|---|
| BDBM50087800 |
|---|
| n/a |
|---|
| Name | BDBM50087800 |
|---|
| Synonyms: | (5-Benzyloxy-1H-indol-2-ylmethyl)-prop-2-ynyl-amine | CHEMBL45069 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H18N2O |
|---|
| Mol. Mass. | 290.359 |
|---|
| SMILES | C#CCNCc1cc2cc(OCc3ccccc3)ccc2[nH]1 |
|---|
| Structure | 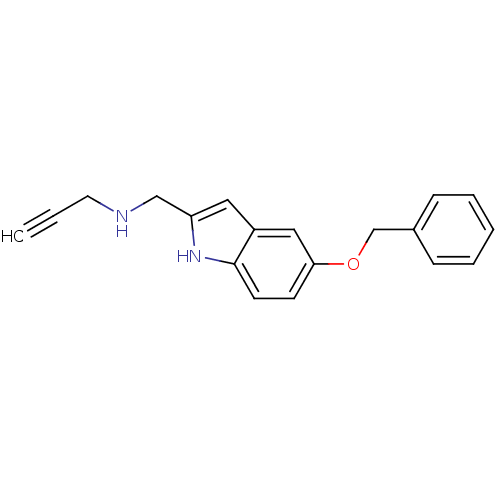
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Tzvetkov, NT; Hinz, S; K�ppers, P; Gastreich, M; M�ller, CE Indazole- and indole-5-carboxamides: selective and reversible monoamine oxidase B inhibitors with subnanomolar potency. J Med Chem57:6679-703 (2014) [PubMed] Article
Tzvetkov, NT; Hinz, S; K�ppers, P; Gastreich, M; M�ller, CE Indazole- and indole-5-carboxamides: selective and reversible monoamine oxidase B inhibitors with subnanomolar potency. J Med Chem57:6679-703 (2014) [PubMed] Article