| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50053305 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1459303 (CHEMBL3367712) |
|---|
| IC50 | 700±n/a nM |
|---|
| Citation |  Laufer, R; Ng, G; Liu, Y; Patel, NK; Edwards, LG; Lang, Y; Li, SW; Feher, M; Awrey, DE; Leung, G; Beletskaya, I; Plotnikova, O; Mason, JM; Hodgson, R; Wei, X; Mao, G; Luo, X; Huang, P; Green, E; Kiarash, R; Lin, DC; Harris-Brandts, M; Ban, F; Nadeem, V; Mak, TW; Pan, GJ; Qiu, W; Chirgadze, NY; Pauls, HW Discovery of inhibitors of the mitotic kinase TTK based on N-(3-(3-sulfamoylphenyl)-1H-indazol-5-yl)-acetamides and carboxamides. Bioorg Med Chem22:4968-97 (2014) [PubMed] Article Laufer, R; Ng, G; Liu, Y; Patel, NK; Edwards, LG; Lang, Y; Li, SW; Feher, M; Awrey, DE; Leung, G; Beletskaya, I; Plotnikova, O; Mason, JM; Hodgson, R; Wei, X; Mao, G; Luo, X; Huang, P; Green, E; Kiarash, R; Lin, DC; Harris-Brandts, M; Ban, F; Nadeem, V; Mak, TW; Pan, GJ; Qiu, W; Chirgadze, NY; Pauls, HW Discovery of inhibitors of the mitotic kinase TTK based on N-(3-(3-sulfamoylphenyl)-1H-indazol-5-yl)-acetamides and carboxamides. Bioorg Med Chem22:4968-97 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50053305 |
|---|
| n/a |
|---|
| Name | BDBM50053305 |
|---|
| Synonyms: | CHEMBL3330409 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H27N5O3S |
|---|
| Mol. Mass. | 477.579 |
|---|
| SMILES | CCc1ccccc1C(N(C)C)C(=O)Nc1ccc2[nH]nc(-c3cccc(c3)S(N)(=O)=O)c2c1 |
|---|
| Structure | 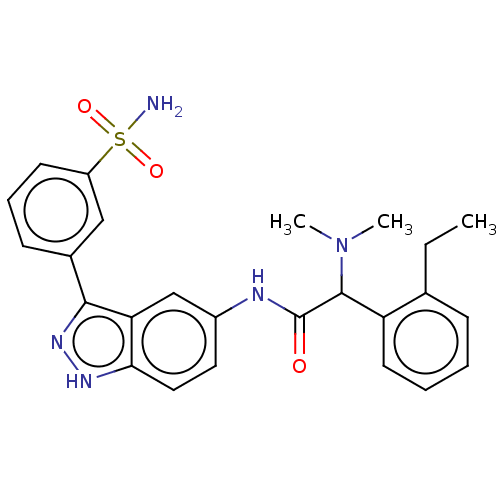
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Laufer, R; Ng, G; Liu, Y; Patel, NK; Edwards, LG; Lang, Y; Li, SW; Feher, M; Awrey, DE; Leung, G; Beletskaya, I; Plotnikova, O; Mason, JM; Hodgson, R; Wei, X; Mao, G; Luo, X; Huang, P; Green, E; Kiarash, R; Lin, DC; Harris-Brandts, M; Ban, F; Nadeem, V; Mak, TW; Pan, GJ; Qiu, W; Chirgadze, NY; Pauls, HW Discovery of inhibitors of the mitotic kinase TTK based on N-(3-(3-sulfamoylphenyl)-1H-indazol-5-yl)-acetamides and carboxamides. Bioorg Med Chem22:4968-97 (2014) [PubMed] Article
Laufer, R; Ng, G; Liu, Y; Patel, NK; Edwards, LG; Lang, Y; Li, SW; Feher, M; Awrey, DE; Leung, G; Beletskaya, I; Plotnikova, O; Mason, JM; Hodgson, R; Wei, X; Mao, G; Luo, X; Huang, P; Green, E; Kiarash, R; Lin, DC; Harris-Brandts, M; Ban, F; Nadeem, V; Mak, TW; Pan, GJ; Qiu, W; Chirgadze, NY; Pauls, HW Discovery of inhibitors of the mitotic kinase TTK based on N-(3-(3-sulfamoylphenyl)-1H-indazol-5-yl)-acetamides and carboxamides. Bioorg Med Chem22:4968-97 (2014) [PubMed] Article