| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histone-lysine N-methyltransferase KMT5C |
|---|
| Ligand | BDBM50063670 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1463680 (CHEMBL3399139) |
|---|
| IC50 | >100000±n/a nM |
|---|
| Citation |  Kung, PP; Huang, B; Zehnder, L; Tatlock, J; Bingham, P; Krivacic, C; Gajiwala, K; Diehl, W; Yu, X; Maegley, KA SAH derived potent and selective EZH2 inhibitors. Bioorg Med Chem Lett25:1532-7 (2015) [PubMed] Article Kung, PP; Huang, B; Zehnder, L; Tatlock, J; Bingham, P; Krivacic, C; Gajiwala, K; Diehl, W; Yu, X; Maegley, KA SAH derived potent and selective EZH2 inhibitors. Bioorg Med Chem Lett25:1532-7 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Histone-lysine N-methyltransferase KMT5C |
|---|
| Name: | Histone-lysine N-methyltransferase KMT5C |
|---|
| Synonyms: | Histone-lysine N-methyltransferase SUV420H2 | KMT5C | KMT5C_HUMAN | Lysine N-methyltransferase 5C | Protein lysine methyltransferase SUV4-20H2 (SUV4-20H2) | SUV420H2 | Su(var)4-20 homolog 2 | Suppressor of variegation 4-20 homolog 2 | Suv4-20h2 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 52147.69 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q86Y97 |
|---|
| Residue: | 462 |
|---|
| Sequence: | MGPDRVTARELCENDDLATSLVLDPYLGFRTHKMNVSPVPPLRRQQHLRSALETFLRQRD
LEAAYRALTLGGWTARYFQSRGPRQEAALKTHVYRYLRAFLPESGFTILPCTRYSMETNG
AKIVSTRAWKKNEKLELLVGCIAELREADEGLLRAGENDFSIMYSTRKRSAQLWLGPAAF
INHDCKPNCKFVPADGNAACVKVLRDIEPGDEVTCFYGEGFFGEKNEHCECHTCERKGEG
AFRTRPREPALPPRPLDKYQLRETKRRLQQGLDSGSRQGLLGPRACVHPSPLRRDPFCAA
CQPLRLPACSARPDTSPLWLQWLPQPQPRVRPRKRRRPRPRRAPVLSTHHAARVSLHRWG
GCGPHCRLRGEALVALGQPPHARWAPQQDWHWARRYGLPYVVRVDLRRLAPAPPATPAPA
GTPGPILIPKQALAFAPFSPPKRLRLVVSHGSIDLDVGGEEL
|
|
|
|---|
| BDBM50063670 |
|---|
| n/a |
|---|
| Name | BDBM50063670 |
|---|
| Synonyms: | CHEMBL3397332 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H29N9O4S |
|---|
| Mol. Mass. | 539.61 |
|---|
| SMILES | N[C@@H](CCSC[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)C(=O)NCc1cc([nH]n1)-c1ccccc1 |r| |
|---|
| Structure | 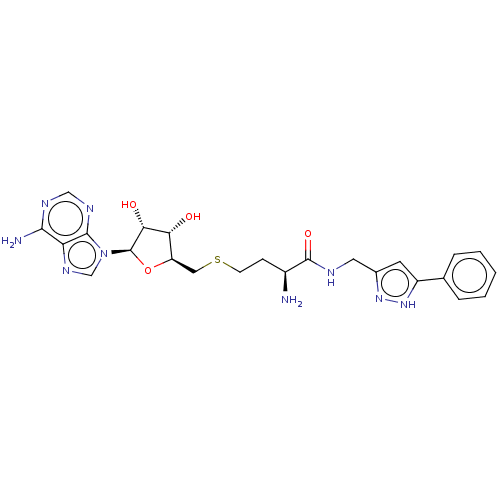
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kung, PP; Huang, B; Zehnder, L; Tatlock, J; Bingham, P; Krivacic, C; Gajiwala, K; Diehl, W; Yu, X; Maegley, KA SAH derived potent and selective EZH2 inhibitors. Bioorg Med Chem Lett25:1532-7 (2015) [PubMed] Article
Kung, PP; Huang, B; Zehnder, L; Tatlock, J; Bingham, P; Krivacic, C; Gajiwala, K; Diehl, W; Yu, X; Maegley, KA SAH derived potent and selective EZH2 inhibitors. Bioorg Med Chem Lett25:1532-7 (2015) [PubMed] Article