| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Urotensin-2 receptor |
|---|
| Ligand | BDBM50089660 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1496221 (CHEMBL3579224) |
|---|
| EC50 | 1.5±n/a nM |
|---|
| Citation |  Dufour-Gallant, J; Chatenet, D; Lubell, WD De Novo Conception of Small Molecule Modulators Based on Endogenous Peptide Ligands: Pyrrolodiazepin-2-oneż-Turn Mimics That Differentially Modulate Urotensin II Receptor-Mediated Vasoconstriction ex Vivo. J Med Chem58:4624-37 (2015) [PubMed] Article Dufour-Gallant, J; Chatenet, D; Lubell, WD De Novo Conception of Small Molecule Modulators Based on Endogenous Peptide Ligands: Pyrrolodiazepin-2-oneż-Turn Mimics That Differentially Modulate Urotensin II Receptor-Mediated Vasoconstriction ex Vivo. J Med Chem58:4624-37 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Urotensin-2 receptor |
|---|
| Name: | Urotensin-2 receptor |
|---|
| Synonyms: | G-protein coupled receptor 14 | G-protein coupled sensory epithelial neuropeptide-like receptor | Gpr14 | Senr | UR-II-R | UR2R_RAT | Urotensin-II | Uts2r |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 42725.34 |
|---|
| Organism: | RAT |
|---|
| Description: | Urotensin-II UTS2R RAT::P49684 |
|---|
| Residue: | 386 |
|---|
| Sequence: | MALSLESTTSFHMLTVSGSTVTELPGDSNVSLNSSWSGPTDPSSLKDLVATGVIGAVLSA
MGVVGMVGNVYTLVVMCRFLRASASMYVYVVNLALADLLYLLSIPFIIATYVTKDWHFGD
VGCRVLFSLDFLTMHASIFTLTIMSSERYAAVLRPLDTVQRSKGYRKLLVLGTWLLALLL
TLPMMLAIQLVRRGSKSLCLPAWGPRAHRTYLTLLFGTSIVGPGLVIGLLYVRLARAYWL
SQQASFKQTRRLPNPRVLYLILGIVLLFWACFLPFWLWQLLAQYHEAMPLTPETARIVNY
LTTCLTYGNSCINPFLYTLLTKNYREYLRGRQRSLGSSCHSPGSPGSFLPSRVHLQQDSG
RSLSSSSQQATETLMLSPVPRNGALL
|
|
|
|---|
| BDBM50089660 |
|---|
| n/a |
|---|
| Name | BDBM50089660 |
|---|
| Synonyms: | CHEMBL3577311 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C30H32N4O4 |
|---|
| Mol. Mass. | 512.5995 |
|---|
| SMILES | NCCCC[C@@H]1N[C@H](c2[nH]c(cc2N(CCc2ccc(O)cc2)C1=O)C(O)=O)c1ccc2ccccc2c1 |r| |
|---|
| Structure | 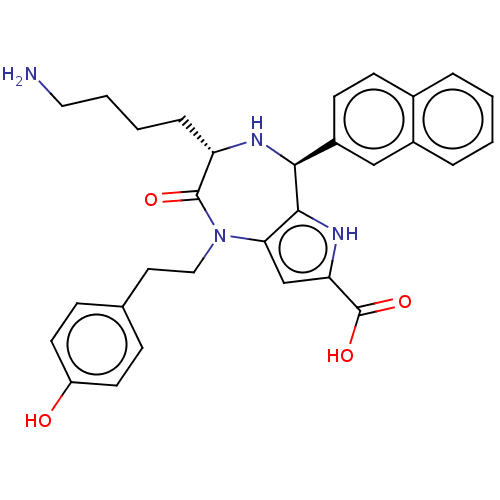
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Dufour-Gallant, J; Chatenet, D; Lubell, WD De Novo Conception of Small Molecule Modulators Based on Endogenous Peptide Ligands: Pyrrolodiazepin-2-oneż-Turn Mimics That Differentially Modulate Urotensin II Receptor-Mediated Vasoconstriction ex Vivo. J Med Chem58:4624-37 (2015) [PubMed] Article
Dufour-Gallant, J; Chatenet, D; Lubell, WD De Novo Conception of Small Molecule Modulators Based on Endogenous Peptide Ligands: Pyrrolodiazepin-2-oneż-Turn Mimics That Differentially Modulate Urotensin II Receptor-Mediated Vasoconstriction ex Vivo. J Med Chem58:4624-37 (2015) [PubMed] Article