| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C9 |
|---|
| Ligand | BDBM50113476 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1510831 (CHEMBL3606595) |
|---|
| IC50 | <15849±n/a nM |
|---|
| Citation |  Christopher, JA; Aves, SJ; Bennett, KA; Dor�, AS; Errey, JC; Jazayeri, A; Marshall, FH; Okrasa, K; Serrano-Vega, MJ; Tehan, BG; Wiggin, GR; Congreve, M Fragment and Structure-Based Drug Discovery for a Class C GPCR: Discovery of the mGlu5 Negative Allosteric Modulator HTL14242 (3-Chloro-5-[6-(5-fluoropyridin-2-yl)pyrimidin-4-yl]benzonitrile). J Med Chem58:6653-64 (2015) [PubMed] Article Christopher, JA; Aves, SJ; Bennett, KA; Dor�, AS; Errey, JC; Jazayeri, A; Marshall, FH; Okrasa, K; Serrano-Vega, MJ; Tehan, BG; Wiggin, GR; Congreve, M Fragment and Structure-Based Drug Discovery for a Class C GPCR: Discovery of the mGlu5 Negative Allosteric Modulator HTL14242 (3-Chloro-5-[6-(5-fluoropyridin-2-yl)pyrimidin-4-yl]benzonitrile). J Med Chem58:6653-64 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C9 |
|---|
| Name: | Cytochrome P450 2C9 |
|---|
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55636.33 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11712 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKV
YGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKW
KEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICS
IIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFM
KSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTE
TTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYID
LLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFK
KSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVP
PFYQLCFIPV
|
|
|
|---|
| BDBM50113476 |
|---|
| n/a |
|---|
| Name | BDBM50113476 |
|---|
| Synonyms: | CHEMBL3603923 | US10246432, Example 17 | US10584111, Ex No. 17 | US11584732, Example 17 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H8ClFN4 |
|---|
| Mol. Mass. | 310.713 |
|---|
| SMILES | Fc1ccc(nc1)-c1cc(ncn1)-c1cc(Cl)cc(c1)C#N |
|---|
| Structure | 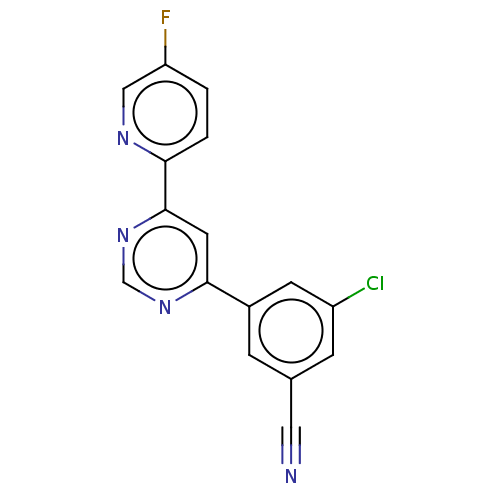
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Christopher, JA; Aves, SJ; Bennett, KA; Dor�, AS; Errey, JC; Jazayeri, A; Marshall, FH; Okrasa, K; Serrano-Vega, MJ; Tehan, BG; Wiggin, GR; Congreve, M Fragment and Structure-Based Drug Discovery for a Class C GPCR: Discovery of the mGlu5 Negative Allosteric Modulator HTL14242 (3-Chloro-5-[6-(5-fluoropyridin-2-yl)pyrimidin-4-yl]benzonitrile). J Med Chem58:6653-64 (2015) [PubMed] Article
Christopher, JA; Aves, SJ; Bennett, KA; Dor�, AS; Errey, JC; Jazayeri, A; Marshall, FH; Okrasa, K; Serrano-Vega, MJ; Tehan, BG; Wiggin, GR; Congreve, M Fragment and Structure-Based Drug Discovery for a Class C GPCR: Discovery of the mGlu5 Negative Allosteric Modulator HTL14242 (3-Chloro-5-[6-(5-fluoropyridin-2-yl)pyrimidin-4-yl]benzonitrile). J Med Chem58:6653-64 (2015) [PubMed] Article