| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Proteinase-activated receptor 1 |
|---|
| Ligand | BDBM50079754 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1539065 (CHEMBL3738281) |
|---|
| IC50 | 52±n/a nM |
|---|
| Citation |  Chao, B; Li, BX; Xiao, X The chemistry and pharmacology of privileged pyrroloquinazolines. Medchemcomm6:510-520 (2015) [PubMed] Chao, B; Li, BX; Xiao, X The chemistry and pharmacology of privileged pyrroloquinazolines. Medchemcomm6:510-520 (2015) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Proteinase-activated receptor 1 |
|---|
| Name: | Proteinase-activated receptor 1 |
|---|
| Synonyms: | CF2R | Coagulation factor II receptor | F2R | PAR-1 | PAR1 | PAR1_HUMAN | Proteinase activated receptor 1 | Proteinase-activated receptor 1 (PAR-1) | TR | Thrombin receptor | Thrombin receptor/ Proteinase-activated receptor 1(Par-1) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 47450.07 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P25116 |
|---|
| Residue: | 425 |
|---|
| Sequence: | MGPRRLLLVAACFSLCGPLLSARTRARRPESKATNATLDPRSFLLRNPNDKYEPFWEDEE
KNESGLTEYRLVSINKSSPLQKQLPAFISEDASGYLTSSWLTLFVPSVYTGVFVVSLPLN
IMAIVVFILKMKVKKPAVVYMLHLATADVLFVSVLPFKISYYFSGSDWQFGSELCRFVTA
AFYCNMYASILLMTVISIDRFLAVVYPMQSLSWRTLGRASFTCLAIWALAIAGVVPLLLK
EQTIQVPGLNITTCHDVLNETLLEGYYAYYFSAFSAVFFFVPLIISTVCYVSIIRCLSSS
AVANRSKKSRALFLSAAVFCIFIICFGPTNVLLIAHYSFLSHTSTTEAAYFAYLLCVCVS
SISCCIDPLIYYYASSECQRYVYSILCCKESSDPSSYNSSGQLMASKMDTCSSNLNNSIY
KKLLT
|
|
|
|---|
| BDBM50079754 |
|---|
| n/a |
|---|
| Name | BDBM50079754 |
|---|
| Synonyms: | CHEMBL65476 | N*3*-Cyclopropyl-7-(4-isopropyl-benzyl)-N*3*-methyl-7H-pyrrolo[3,2-f]quinazoline-1,3-diamine |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H27N5 |
|---|
| Mol. Mass. | 385.5047 |
|---|
| SMILES | CC(C)c1ccc(Cn2ccc3c2ccc2nc(nc(N)c32)N(C)C2CC2)cc1 |
|---|
| Structure | 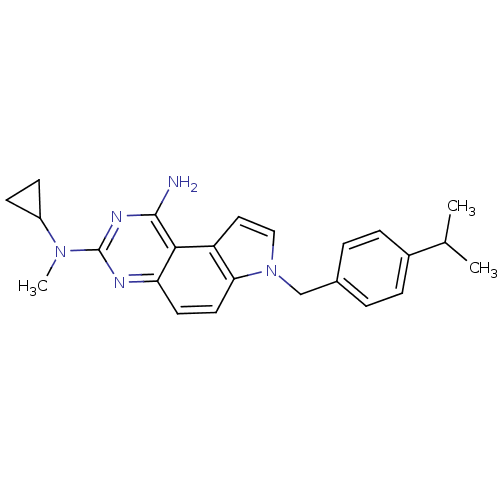
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Chao, B; Li, BX; Xiao, X The chemistry and pharmacology of privileged pyrroloquinazolines. Medchemcomm6:510-520 (2015) [PubMed]
Chao, B; Li, BX; Xiao, X The chemistry and pharmacology of privileged pyrroloquinazolines. Medchemcomm6:510-520 (2015) [PubMed]