| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-1D adrenergic receptor |
|---|
| Ligand | BDBM50164737 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1575788 (CHEMBL3801620) |
|---|
| Ki | 160±n/a nM |
|---|
| Citation |  Sakauchi, N; Kohara, Y; Sato, A; Suzaki, T; Imai, Y; Okabe, Y; Imai, S; Saikawa, R; Nagabukuro, H; Kuno, H; Fujita, H; Kamo, I; Yoshida, M Discovery of 5-Chloro-1-(5-chloro-2-(methylsulfonyl)benzyl)-2-imino-1,2-dihydropyridine-3-carboxamide (TAK-259) as a Novel, Selective, and Orally Activea1D Adrenoceptor Antagonist with Antiurinary Frequency Effects: Reducing Human Ether-a-go-go-Related Gene (hERG) Liabilities. J Med Chem59:2989-3002 (2016) [PubMed] Article Sakauchi, N; Kohara, Y; Sato, A; Suzaki, T; Imai, Y; Okabe, Y; Imai, S; Saikawa, R; Nagabukuro, H; Kuno, H; Fujita, H; Kamo, I; Yoshida, M Discovery of 5-Chloro-1-(5-chloro-2-(methylsulfonyl)benzyl)-2-imino-1,2-dihydropyridine-3-carboxamide (TAK-259) as a Novel, Selective, and Orally Activea1D Adrenoceptor Antagonist with Antiurinary Frequency Effects: Reducing Human Ether-a-go-go-Related Gene (hERG) Liabilities. J Med Chem59:2989-3002 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-1D adrenergic receptor |
|---|
| Name: | Alpha-1D adrenergic receptor |
|---|
| Synonyms: | ADA1D_HUMAN | ADRA1A | ADRA1D | Adrenergic receptor | Adrenergic receptor alpha | Alpha 1D-adrenoceptor | Alpha 1D-adrenoreceptor | Alpha adrenergic receptor (1a and 1d) | Alpha-1D adrenoceptor | Alpha-adrenergic receptor 1a | adrenergic Alpha1D |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 60485.82 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | adrenergic Alpha1D ADRA1D HUMAN::P25100 |
|---|
| Residue: | 572 |
|---|
| Sequence: | MTFRDLLSVSFEGPRPDSSAGGSSAGGGGGSAGGAAPSEGPAVGGVPGGAGGGGGVVGAG
SGEDNRSSAGEPGSAGAGGDVNGTAAVGGLVVSAQGVGVGVFLAAFILMAVAGNLLVILS
VACNRHLQTVTNYFIVNLAVADLLLSATVLPFSATMEVLGFWAFGRAFCDVWAAVDVLCC
TASILSLCTISVDRYVGVRHSLKYPAIMTERKAAAILALLWVVALVVSVGPLLGWKEPVP
PDERFCGITEEAGYAVFSSVCSFYLPMAVIVVMYCRVYVVARSTTRSLEAGVKRERGKAS
EVVLRIHCRGAATGADGAHGMRSAKGHTFRSSLSVRLLKFSREKKAAKTLAIVVGVFVLC
WFPFFFVLPLGSLFPQLKPSEGVFKVIFWLGYFNSCVNPLIYPCSSREFKRAFLRLLRCQ
CRRRRRRRPLWRVYGHHWRASTSGLRQDCAPSSGDAPPGAPLALTALPDPDPEPPGTPEM
QAPVASRRKPPSAFREWRLLGPFRRPTTQLRAKVSSLSHKIRAGGAQRAEAACAQRSEVE
AVSLGVPHEVAEGATCQAYELADYSNLRETDI
|
|
|
|---|
| BDBM50164737 |
|---|
| n/a |
|---|
| Name | BDBM50164737 |
|---|
| Synonyms: | CHEMBL3797500 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C13H12BrN3O |
|---|
| Mol. Mass. | 306.158 |
|---|
| SMILES | NC(=O)c1cc(Br)cn(Cc2ccccc2)c1=N |
|---|
| Structure | 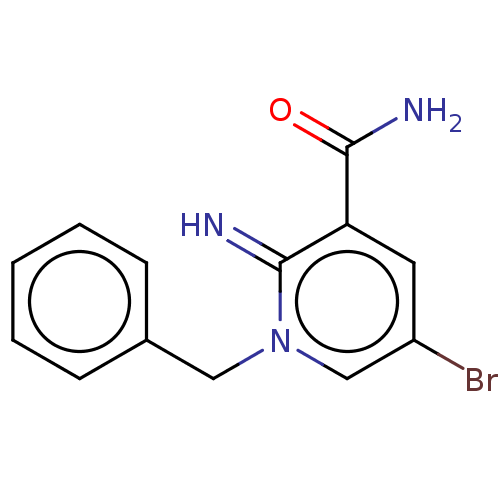
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Sakauchi, N; Kohara, Y; Sato, A; Suzaki, T; Imai, Y; Okabe, Y; Imai, S; Saikawa, R; Nagabukuro, H; Kuno, H; Fujita, H; Kamo, I; Yoshida, M Discovery of 5-Chloro-1-(5-chloro-2-(methylsulfonyl)benzyl)-2-imino-1,2-dihydropyridine-3-carboxamide (TAK-259) as a Novel, Selective, and Orally Activea1D Adrenoceptor Antagonist with Antiurinary Frequency Effects: Reducing Human Ether-a-go-go-Related Gene (hERG) Liabilities. J Med Chem59:2989-3002 (2016) [PubMed] Article
Sakauchi, N; Kohara, Y; Sato, A; Suzaki, T; Imai, Y; Okabe, Y; Imai, S; Saikawa, R; Nagabukuro, H; Kuno, H; Fujita, H; Kamo, I; Yoshida, M Discovery of 5-Chloro-1-(5-chloro-2-(methylsulfonyl)benzyl)-2-imino-1,2-dihydropyridine-3-carboxamide (TAK-259) as a Novel, Selective, and Orally Activea1D Adrenoceptor Antagonist with Antiurinary Frequency Effects: Reducing Human Ether-a-go-go-Related Gene (hERG) Liabilities. J Med Chem59:2989-3002 (2016) [PubMed] Article