| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Acetylcholinesterase |
|---|
| Ligand | BDBM8963 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1615950 (CHEMBL3858019) |
|---|
| IC50 | 5.3±n/a nM |
|---|
| Citation |  Ceschi, MA; da Costa, JS; Lopes, JPB; C�mara, VS; Campo, LF; Borges, ACA; Gon�alves, CAS; de Souza, DF; Konrath, EL; Karl, ALM; Guedes, IA; Dardenne, LE Novel series of tacrine-tianeptine hybrids: Synthesis, cholinesterase inhibitory activity, S100B secretion and a molecular modeling approach. Eur J Med Chem121:758-772 (2016) [PubMed] Article Ceschi, MA; da Costa, JS; Lopes, JPB; C�mara, VS; Campo, LF; Borges, ACA; Gon�alves, CAS; de Souza, DF; Konrath, EL; Karl, ALM; Guedes, IA; Dardenne, LE Novel series of tacrine-tianeptine hybrids: Synthesis, cholinesterase inhibitory activity, S100B secretion and a molecular modeling approach. Eur J Med Chem121:758-772 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Acetylcholinesterase |
|---|
| Name: | Acetylcholinesterase |
|---|
| Synonyms: | ACES_RAT | Acetylcholinesterase (AChE) | Acetylcholinesterase and butyrylcholinesterase (AChE and BChE) | Acetylcholinesterase precursor | Acetylcholinesterase, AChE | Ache |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 68193.62 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | P37136 |
|---|
| Residue: | 614 |
|---|
| Sequence: | MRPPWYPLHTPSLASPLLFLLLSLLGGGARAEGREDPQLLVRVRGGQLRGIRLKAPGGPV
SAFLGIPFAEPPVGSRRFMPPEPKRPWSGILDATTFQNVCYQYVDTLYPGFEGTEMWNPN
RELSEDCLYLNVWTPYPRPTSPTPVLIWIYGGGFYSGASSLDVYDGRFLAQVEGTVLVSM
NYRVGTFGFLALPGSREAPGNVGLLDQRLALQWVQENIAAFGGDPMSVTLFGESAGAASV
GMHILSLPSRSLFHRAVLQSGTPNGPWATVSAGEARRRATLLARLVGCPPGGAGGNDTEL
ISCLRTRPAQDLVDHEWHVLPQESIFRFSFVPVVDGDFLSDTPDALINTGDFQDLQVLVG
VVKDEGSYFLVYGVPGFSKDNESLISRAQFLAGVRIGVPQASDLAAEAVVLHYTDWLHPE
DPAHLRDAMSAVVGDHNVVCPVAQLAGRLAAQGARVYAYIFEHRASTLTWPLWMGVPHGY
EIEFIFGLPLDPSLNYTVEERIFAQRLMQYWTNFARTGDPNDPRDSKSPRWPPYTTAAQQ
YVSLNLKPLEVRRGLRAQTCAFWNRFLPKLLSATDTLDEAERQWKAEFHRWSSYMVHWKN
QFDHYSKQERCSDL
|
|
|
|---|
| BDBM8963 |
|---|
| n/a |
|---|
| Name | BDBM8963 |
|---|
| Synonyms: | CHEMBL32823 | Homodimeric Tacrine Analog 3b | N-[7-(1,2,3,4-tetrahydroacridin-9-ylamino)heptyl]-1,2,3,4-tetrahydroacridin-9-amine | Tacrine Dimer 3b | Tacrine-Based Inhibitor 2f | tacrine homobivalent compound 3a |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C33H40N4 |
|---|
| Mol. Mass. | 492.6975 |
|---|
| SMILES | C(CCCNc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 |
|---|
| Structure | 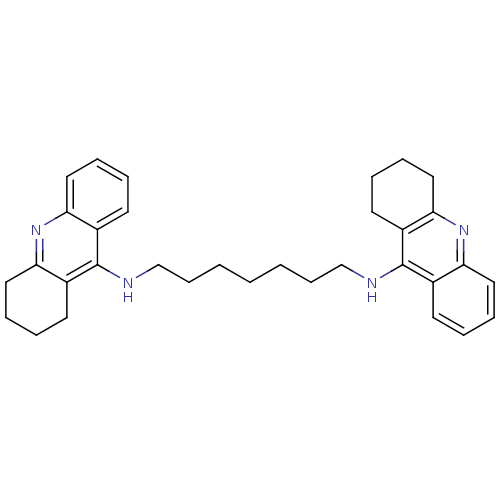
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ceschi, MA; da Costa, JS; Lopes, JPB; C�mara, VS; Campo, LF; Borges, ACA; Gon�alves, CAS; de Souza, DF; Konrath, EL; Karl, ALM; Guedes, IA; Dardenne, LE Novel series of tacrine-tianeptine hybrids: Synthesis, cholinesterase inhibitory activity, S100B secretion and a molecular modeling approach. Eur J Med Chem121:758-772 (2016) [PubMed] Article
Ceschi, MA; da Costa, JS; Lopes, JPB; C�mara, VS; Campo, LF; Borges, ACA; Gon�alves, CAS; de Souza, DF; Konrath, EL; Karl, ALM; Guedes, IA; Dardenne, LE Novel series of tacrine-tianeptine hybrids: Synthesis, cholinesterase inhibitory activity, S100B secretion and a molecular modeling approach. Eur J Med Chem121:758-772 (2016) [PubMed] Article