| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 11B2, mitochondrial |
|---|
| Ligand | BDBM50206963 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1629493 (CHEMBL3872119) |
|---|
| IC50 | 0.600000±n/a nM |
|---|
| Citation |  Whitehead, BR; Lo, MM; Ali, A; Park, MK; Hoyt, SB; Xiong, Y; Cai, J; Carswell, E; Cooke, A; MacLean, J; Ratcliffe, P; Robinson, J; Bennett, DJ; Clemas, JA; Wisniewski, T; Struthers, M; Cully, D; MacNeil, DJ Imidazopyridyl compounds as aldosterone synthase inhibitors. Bioorg Med Chem Lett27:143-146 (2017) [PubMed] Article Whitehead, BR; Lo, MM; Ali, A; Park, MK; Hoyt, SB; Xiong, Y; Cai, J; Carswell, E; Cooke, A; MacLean, J; Ratcliffe, P; Robinson, J; Bennett, DJ; Clemas, JA; Wisniewski, T; Struthers, M; Cully, D; MacNeil, DJ Imidazopyridyl compounds as aldosterone synthase inhibitors. Bioorg Med Chem Lett27:143-146 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 11B2, mitochondrial |
|---|
| Name: | Cytochrome P450 11B2, mitochondrial |
|---|
| Synonyms: | Aldosterone Synthase (CYP11B2) | Aldosterone synthase | Aldosterone-synthesizing enzyme | C11B2_HUMAN | CYP11B2 | CYPXIB2 | Cytochrome P450 11B2 | Cytochrome P450 11B2 (CYP11B2) | Cytochrome P450 11B2, mitochondrial | P-450Aldo | P-450C18 | Steroid 18-hydroxylase |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 57582.15 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P19099 |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALRAKAEVCVAAPWLSLQRARALGTRAARAPRTVLPFEAMPQHPGNRWLRLLQIWREQG
YEHLHLEMHQTFQELGPIFRYNLGGPRMVCVMLPEDVEKLQQVDSLHPCRMILEPWVAYR
QHRGHKCGVFLLNGPEWRFNRLRLNPDVLSPKAVQRFLPMVDAVARDFSQALKKKVLQNA
RGSLTLDVQPSIFHYTIEASNLALFGERLGLVGHSPSSASLNFLHALEVMFKSTVQLMFM
PRSLSRWISPKVWKEHFEAWDCIFQYGDNCIQKIYQELAFNRPQHYTGIVAELLLKAELS
LEAIKANSMELTAGSVDTTAFPLLMTLFELARNPDVQQILRQESLAAAASISEHPQKATT
ELPLLRAALKETLRLYPVGLFLERVVSSDLVLQNYHIPAGTLVQVFLYSLGRNAALFPRP
ERYNPQRWLDIRGSGRNFHHVPFGFGMRQCLGRRLAEAEMLLLLHHVLKHFLVETLTQED
IKMVYSFILRPGTSPLLTFRAIN
|
|
|
|---|
| BDBM50206963 |
|---|
| n/a |
|---|
| Name | BDBM50206963 |
|---|
| Synonyms: | CHEMBL3948337 | US9518055, Example 41 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H14FN3 |
|---|
| Mol. Mass. | 303.333 |
|---|
| SMILES | Fc1ccc2nc(c(C3CC3)n2c1)-c1cncc2ccccc12 |
|---|
| Structure | 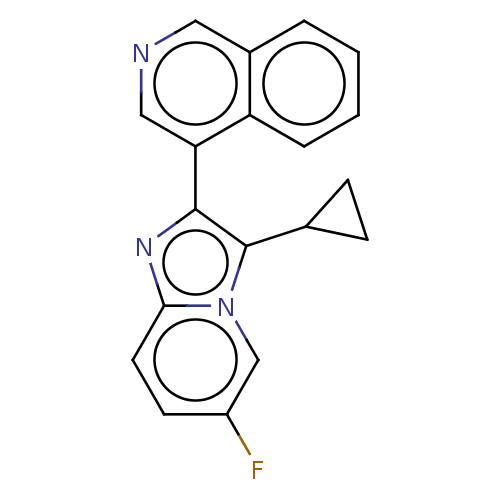
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Whitehead, BR; Lo, MM; Ali, A; Park, MK; Hoyt, SB; Xiong, Y; Cai, J; Carswell, E; Cooke, A; MacLean, J; Ratcliffe, P; Robinson, J; Bennett, DJ; Clemas, JA; Wisniewski, T; Struthers, M; Cully, D; MacNeil, DJ Imidazopyridyl compounds as aldosterone synthase inhibitors. Bioorg Med Chem Lett27:143-146 (2017) [PubMed] Article
Whitehead, BR; Lo, MM; Ali, A; Park, MK; Hoyt, SB; Xiong, Y; Cai, J; Carswell, E; Cooke, A; MacLean, J; Ratcliffe, P; Robinson, J; Bennett, DJ; Clemas, JA; Wisniewski, T; Struthers, M; Cully, D; MacNeil, DJ Imidazopyridyl compounds as aldosterone synthase inhibitors. Bioorg Med Chem Lett27:143-146 (2017) [PubMed] Article