| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 1B |
|---|
| Ligand | BDBM50219085 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1374 (CHEMBL616447) |
|---|
| Ki | 1259±n/a nM |
|---|
| Citation |  Bromidge, SM; Griffith, K; Heightman, TD; Jennings, A; King, FD; Moss, SF; Newman, H; Riley, G; Routledge, C; Serafinowska, HT; Thomas, DR Novel (4-piperazin-1-ylquinolin-6-yl) arylsulfonamides with high affinity and selectivity for the 5-HT(6) receptor. Bioorg Med Chem Lett11:2843-6 (2001) [PubMed] Bromidge, SM; Griffith, K; Heightman, TD; Jennings, A; King, FD; Moss, SF; Newman, H; Riley, G; Routledge, C; Serafinowska, HT; Thomas, DR Novel (4-piperazin-1-ylquinolin-6-yl) arylsulfonamides with high affinity and selectivity for the 5-HT(6) receptor. Bioorg Med Chem Lett11:2843-6 (2001) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 1B |
|---|
| Name: | 5-hydroxytryptamine receptor 1B |
|---|
| Synonyms: | 5-HT-1B | 5-HT-1D-beta | 5-HT1B | 5-hydroxytryptamine receptor 1B (5-HT1B) | 5HT1B_HUMAN | HTR1B | HTR1DB | S12 | Serotonin (5-HT) receptor | Serotonin 1D beta receptor | Serotonin Receptor 1B |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 43579.17 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Receptor binding assays were performed using human clone stably expressed in CHO cells |
|---|
| Residue: | 390 |
|---|
| Sequence: | MEEPGAQCAPPPPAGSETWVPQANLSSAPSQNCSAKDYIYQDSISLPWKVLLVMLLALIT
LATTLSNAFVIATVYRTRKLHTPANYLIASLAVTDLLVSILVMPISTMYTVTGRWTLGQV
VCDFWLSSDITCCTASILHLCVIALDRYWAITDAVEYSAKRTPKRAAVMIALVWVFSISI
SLPPFFWRQAKAEEEVSECVVNTDHILYTVYSTVGAFYFPTLLLIALYGRIYVEARSRIL
KQTPNRTGKRLTRAQLITDSPGSTSSVTSINSRVPDVPSESGSPVYVNQVKVRVSDALLE
KKKLMAARERKATKTLGIILGAFIVCWLPFFIISLVMPICKDACWFHLAIFDFFTWLGYL
NSLINPIIYTMSNEDFKQAFHKLIRFKCTS
|
|
|
|---|
| BDBM50219085 |
|---|
| n/a |
|---|
| Name | BDBM50219085 |
|---|
| Synonyms: | CHEMBL97211 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H23ClN4O2S2 |
|---|
| Mol. Mass. | 487.037 |
|---|
| SMILES | Cc1c(sc2ccc(Cl)cc12)S(=O)(=O)Nc1ccc2nc(C)cc(N3CCNCC3)c2c1 |
|---|
| Structure | 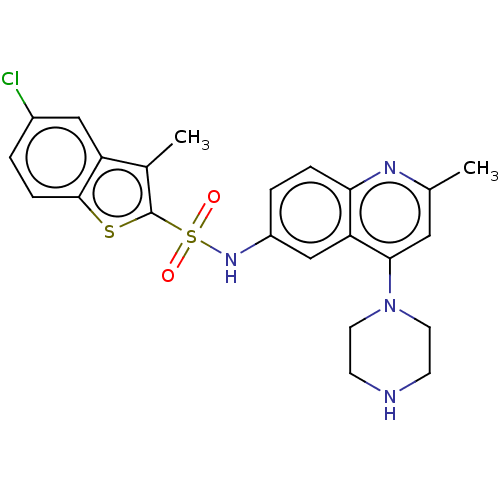
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bromidge, SM; Griffith, K; Heightman, TD; Jennings, A; King, FD; Moss, SF; Newman, H; Riley, G; Routledge, C; Serafinowska, HT; Thomas, DR Novel (4-piperazin-1-ylquinolin-6-yl) arylsulfonamides with high affinity and selectivity for the 5-HT(6) receptor. Bioorg Med Chem Lett11:2843-6 (2001) [PubMed]
Bromidge, SM; Griffith, K; Heightman, TD; Jennings, A; King, FD; Moss, SF; Newman, H; Riley, G; Routledge, C; Serafinowska, HT; Thomas, DR Novel (4-piperazin-1-ylquinolin-6-yl) arylsulfonamides with high affinity and selectivity for the 5-HT(6) receptor. Bioorg Med Chem Lett11:2843-6 (2001) [PubMed]