| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cholecystokinin receptor type A/Gastrin/cholecystokinin type B receptor |
|---|
| Ligand | BDBM50016425 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_49554 (CHEMBL663470) |
|---|
| IC50 | 1.5±n/a nM |
|---|
| Citation |  Rodriguez, M; Galas, MC; Lignon, MF; Mendre, C; Laur, J; Aumelas, A; Martinez, J Synthesis and biological activity of some partially modified retro-inverso analogues of cholecystokinin. J Med Chem32:2331-9 (1989) [PubMed] Rodriguez, M; Galas, MC; Lignon, MF; Mendre, C; Laur, J; Aumelas, A; Martinez, J Synthesis and biological activity of some partially modified retro-inverso analogues of cholecystokinin. J Med Chem32:2331-9 (1989) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cholecystokinin receptor type A/Gastrin/cholecystokinin type B receptor |
|---|
| Name: | Cholecystokinin receptor type A/Gastrin/cholecystokinin type B receptor |
|---|
| Synonyms: | Cholecystokinin receptor |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | n/a |
|---|
| Description: | ASSAY_ID of ChEMBL is 1544143 |
|---|
| Components: | This complex has 2 components. |
|---|
| Component 1 |
| Name: | Gastrin/cholecystokinin type B receptor |
|---|
| Synonyms: | Cckbr | Cholecystokinin A | Cholecystokinin B receptor | Cholecystokinin receptor | GASR_RAT | Gastrin/cholecystokinin type B receptor |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 48980.43 |
|---|
| Organism: | RAT |
|---|
| Description: | Cholecystokinin A CCKBR RAT::P30553 |
|---|
| Residue: | 452 |
|---|
| Sequence: | MELLKLNRSVQGPGPGSGSSLCRPGVSLLNSSSAGNLSCDPPRIRGTGTRELEMAIRITL
YAVIFLMSVGGNVLIIVVLGLSRRLRTVTNAFLLSLAVSDLLLAVACMPFTLLPNLMGTF
IFGTVICKAISYLMGVSVSVSTLNLVAIALERYSAICRPLQARVWQTRSHAARVILATWL
LSGLLMVPYPVYTMVQPVGPRVLQCMHRWPSARVQQTWSVLLLLLLFFIPGVVIAVAYGL
ISRELYLGLHFDGENDSETQSRARNQGGLPGGAAPGPVHQNGGCRPVTSVAGEDSDGCCV
QLPRSRLEMTTLTTPTPGPVPGPRPNQAKLLAKKRVVRMLLVIVLLFFLCWLPVYSVNTW
RAFDGPGAQRALSGAPISFIHLLSYVSACVNPLVYCFMHRRFRQACLDTCARCCPRPPRA
RPQPLPDEDPPTPSIASLSRLSYTTISTLGPG
|
|
|
|---|
| Component 2 |
| Name: | Cholecystokinin receptor type A |
|---|
| Synonyms: | CCKAR_RAT | Cckar | Cholecystokinin peripheral | Cholecystokinin receptor | Cholecystokinin receptor type A |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 49676.37 |
|---|
| Organism: | RAT |
|---|
| Description: | Cholecystokinin central 0 RAT::P30551 |
|---|
| Residue: | 444 |
|---|
| Sequence: | MSHSPARQHLVESSRMDVVDSLLMNGSNITPPCELGLENETLFCLDQPQPSKEWQSALQI
LLYSIIFLLSVLGNTLVITVLIRNKRMRTVTNIFLLSLAVSDLMLCLFCMPFNLIPNLLK
DFIFGSAVCKTTTYFMGTSVSVSTFNLVAISLERYGAICRPLQSRVWQTKSHALKVIAAT
WCLSFTIMTPYPIYSNLVPFTKNNNQTANMCRFLLPSDAMQQSWQTFLLLILFLLPGIVM
VVAYGLISLELYQGIKFDASQKKSAKEKKPSTGSSTRYEDSDGCYLQKSRPPRKLELQQL
SSGSGGSRLNRIRSSSSAANLIAKKRVIRMLIVIVVLFFLCWMPIFSANAWRAYDTVSAE
KHLSGTPISFILLLSYTSSCVNPIIYCFMNKRFRLGFMATFPCCPNPGPPGVRGEVGEEE
DGRTIRALLSRYSYSHMSTSAPPP
|
|
|
|---|
| BDBM50016425 |
|---|
| n/a |
|---|
| Name | BDBM50016425 |
|---|
| Synonyms: | (S)-3-{(S)-2-[(S)-2-(2-{(S)-2-[(S)-2-tert-Butoxycarbonylamino-3-(4-sulfooxy-phenyl)-propionylamino]-hexanoylamino}-acetylamino)-3-(1H-indol-3-yl)-propionylamino]-hexanoylamino}-N-((S)-1-carbamoyl-2-phenyl-ethyl)-succinamic acid | 3-{2-[2-(2-{2-[2-(tert-Butoxycarbonyl-methyl-amino)-3-(4-sulfonyl-oxy-phenyl)-propionylamino]-hexanoylamino}-acetylamino)-3-(1H-indol-3-yl)-propionylamino]-hexanoylamino}-N-(1-carbamoyl-2-phenyl-ethyl)-succinamic acid | 3-{2-[2-(2-{2-[2-tert-Butoxycarbonylamino-3-(4-sulfooxy-phenyl)-propionylamino]-hexanoylamino}-acetylamino)-3-(1H-indol-3-yl)-propionylamino]-hexanoylamino}-N-(1-carbamoyl-2-phenyl-ethyl)-succinamic acid | Boc-Tyr(SO3H)-Nle-Gly-Trp-Nle-Asp-Phe-NH2 | CHEMBL384035 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C52H69N9O15S |
|---|
| Mol. Mass. | 1092.221 |
|---|
| SMILES | CCCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)OC(C)(C)C)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCCC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |
|---|
| Structure | 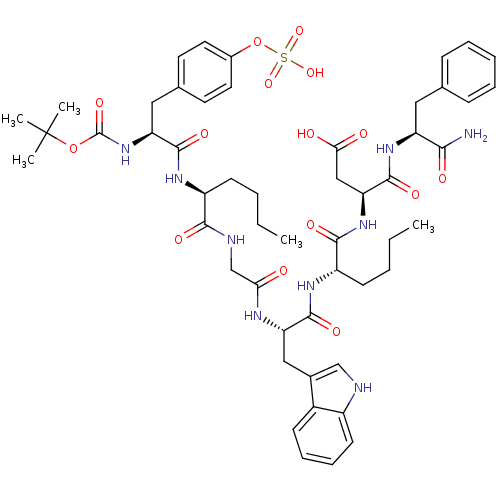
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Rodriguez, M; Galas, MC; Lignon, MF; Mendre, C; Laur, J; Aumelas, A; Martinez, J Synthesis and biological activity of some partially modified retro-inverso analogues of cholecystokinin. J Med Chem32:2331-9 (1989) [PubMed]
Rodriguez, M; Galas, MC; Lignon, MF; Mendre, C; Laur, J; Aumelas, A; Martinez, J Synthesis and biological activity of some partially modified retro-inverso analogues of cholecystokinin. J Med Chem32:2331-9 (1989) [PubMed]