| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2A |
|---|
| Ligand | BDBM50064708 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1660349 (CHEMBL4009961) |
|---|
| Ki | 0.200000±n/a nM |
|---|
| Citation |  Glennon, RA The 2014 Philip S. Portoghese Medicinal Chemistry Lectureship: The"Phenylalkylaminome" with a Focus on Selected Drugs of Abuse. J Med Chem60:2605-2628 (2017) [PubMed] Article Glennon, RA The 2014 Philip S. Portoghese Medicinal Chemistry Lectureship: The"Phenylalkylaminome" with a Focus on Selected Drugs of Abuse. J Med Chem60:2605-2628 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2A |
|---|
| Name: | 5-hydroxytryptamine receptor 2A |
|---|
| Synonyms: | 5-HT-2A | 5-HT2 | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_RAT | Htr2 | Htr2a | Serotonin Receptor 2A |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 52852.05 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Rat cortex membranes 5-HT2A receptors. |
|---|
| Residue: | 471 |
|---|
| Sequence: | MEILCEDNISLSSIPNSLMQLGDGPRLYHNDFNSRDANTSEASNWTIDAENRTNLSCEGY
LPPTCLSILHLQEKNWSALLTTVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIAD
MLLGFLVMPVSMLTILYGYRWPLPSKLCAIWIYLDVLFSTASIMHLCAISLDRYVAIQNP
IHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSF
VAFFIPLTIMVITYFLTIKSLQKEATLCVSDLSTRAKLASFSFLPQSSLSSEKLFQRSIH
REPGSYAGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNENVIGA
LLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENRKPLQLILVNTIPALAYK
SSQLQVGQKKNSQEDAEQTVDDCSMVTLGKQQSEENCTDNIETVNEKVSCV
|
|
|
|---|
| BDBM50064708 |
|---|
| n/a |
|---|
| Name | BDBM50064708 |
|---|
| Synonyms: | (R)-1-(4-bromo-2,5-dimethoxyphenyl)propan-2-amine | (R)-2-(4-Bromo-2,5-dimethoxy-phenyl)-1-methyl-ethylamine | CHEMBL69700 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C11H16BrNO2 |
|---|
| Mol. Mass. | 274.154 |
|---|
| SMILES | COc1cc(C[C@@H](C)N)c(OC)cc1Br |r| |
|---|
| Structure | 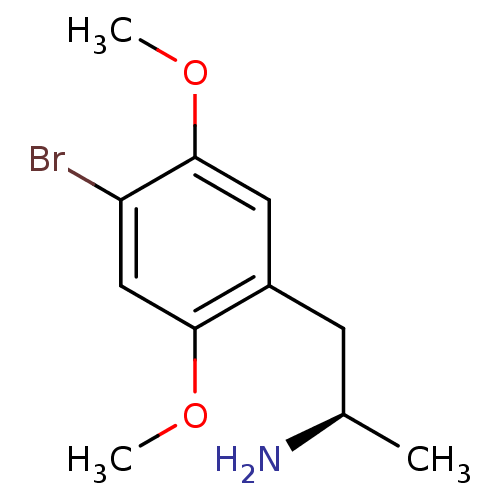
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Glennon, RA The 2014 Philip S. Portoghese Medicinal Chemistry Lectureship: The"Phenylalkylaminome" with a Focus on Selected Drugs of Abuse. J Med Chem60:2605-2628 (2017) [PubMed] Article
Glennon, RA The 2014 Philip S. Portoghese Medicinal Chemistry Lectureship: The"Phenylalkylaminome" with a Focus on Selected Drugs of Abuse. J Med Chem60:2605-2628 (2017) [PubMed] Article