| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Nociceptin receptor |
|---|
| Ligand | BDBM21842 |
|---|
| Substrate/Competitor | n/a |
|---|
| Ki | 0.38±n/a nM |
|---|
| Comments | PDSP_3168 |
|---|
| Citation |  Neal, CR; Owens, CE; Taylor, LP; Hoversten, MT; Akil, H; Watson, SJ Binding and GTPgammaS autoradiographic analysis of preproorphanin precursor peptide products at the ORL1 and opioid receptors. J Chem Neuroanat25:233-47 (2003) [PubMed] Article Neal, CR; Owens, CE; Taylor, LP; Hoversten, MT; Akil, H; Watson, SJ Binding and GTPgammaS autoradiographic analysis of preproorphanin precursor peptide products at the ORL1 and opioid receptors. J Chem Neuroanat25:233-47 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article |
|---|
| |
| Nociceptin receptor |
|---|
| Name: | Nociceptin receptor |
|---|
| Synonyms: | Nociceptin/Orphanin FQ, NOP receptor | Nociceptin/mu opioid receptor | OPRX_RAT | Oor | Oprl | Oprl1 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 40531.08 |
|---|
| Organism: | RAT |
|---|
| Description: | Nociceptin/Orphanin FQ, NOP receptor 0 RAT::P35370 |
|---|
| Residue: | 367 |
|---|
| Sequence: | MESLFPAPYWEVLYGSHFQGNLSLLNETVPHHLLLNASHSAFLPLGLKVTIVGLYLAVCI
GGLLGNCLVMYVILRHTKMKTATNIYIFNLALADTLVLLTLPFQGTDILLGFWPFGNALC
KTVIAIDYYNMFTSTFTLTAMSVDRYVAICHPIRALDVRTSSKAQAVNVAIWALASVVGV
PVAIMGSAQVEDEEIECLVEIPAPQDYWGPVFAICIFLFSFIIPVLIISVCYSLMIRRLR
GVRLLSGSREKDRNLRRITRLVLVVVAVFVGCWTPVQVFVLVQGLGVQPGSETAVAILRF
CTALGYVNSCLNPILYAFLDENFKACFRKFCCASSLHREMQVSDRVRSIAKDVGLGCKTS
ETVPRPA
|
|
|
|---|
| BDBM21842 |
|---|
| n/a |
|---|
| Name | BDBM21842 |
|---|
| Synonyms: | (2S)-2-[(2S)-2-[(2S)-2-[(2S)-2-[(2S)-6-amino-2-[(2S)-2-[(2S)-2-[(2S)-2-[(2S)-6-amino-2-[(2S)-2-[(2S)-2-{2-[(2S,3R)-2-[(2S)-2-(2-{2-[(2S)-2-amino-3-phenylpropanamido]acetamido}acetamido)-3-phenylpropanamido]-3-hydroxybutanamido]acetamido}propanamido]-5-carbamimidamidopentanamido]hexanamido]-3-hydroxypropanamido]propanamido]-5-carbamimidamidopentanamido]hexanamido]-4-methylpentanamido]propanamido]butanediamido]-4-carbamoylbutanoic acid | N/OFQ | OFQ 1-17 | OFQ/N | Orphanin FQ | [125I]-nociceptin | nociceptin |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C79H129N27O22 |
|---|
| Mol. Mass. | 1809.0373 |
|---|
| SMILES | [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#8])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccccc1)-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@@H](-[#7])-[#6]-c1ccccc1)-[#6@@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6](-[#7])=O)-[#6](-[#8])=O |
|---|
| Structure | 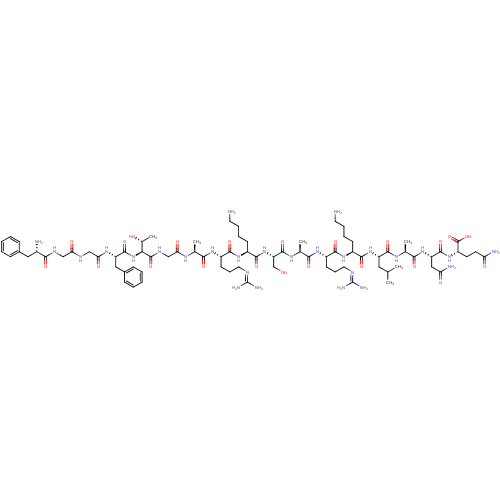
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Neal, CR; Owens, CE; Taylor, LP; Hoversten, MT; Akil, H; Watson, SJ Binding and GTPgammaS autoradiographic analysis of preproorphanin precursor peptide products at the ORL1 and opioid receptors. J Chem Neuroanat25:233-47 (2003) [PubMed] Article
Neal, CR; Owens, CE; Taylor, LP; Hoversten, MT; Akil, H; Watson, SJ Binding and GTPgammaS autoradiographic analysis of preproorphanin precursor peptide products at the ORL1 and opioid receptors. J Chem Neuroanat25:233-47 (2003) [PubMed] Article