| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Muscarinic acetylcholine receptor M4 |
|---|
| Ligand | BDBM50048803 |
|---|
| Substrate/Competitor | n/a |
|---|
| Ki | 1600±n/a nM |
|---|
| Comments | PDSP_1996 |
|---|
| Citation |  Bymaster, FP; Felder, CC; Tzavara, E; Nomikos, GG; Calligaro, DO; Mckinzie, DL Muscarinic mechanisms of antipsychotic atypicality. Prog Neuropsychopharmacol Biol Psychiatry27:1125-43 (2003) [PubMed] Article Bymaster, FP; Felder, CC; Tzavara, E; Nomikos, GG; Calligaro, DO; Mckinzie, DL Muscarinic mechanisms of antipsychotic atypicality. Prog Neuropsychopharmacol Biol Psychiatry27:1125-43 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article |
|---|
| |
| Muscarinic acetylcholine receptor M4 |
|---|
| Name: | Muscarinic acetylcholine receptor M4 |
|---|
| Synonyms: | ACM4_HUMAN | CHRM4 | Cholinergic, muscarinic M4 | Muscarinic acetylcholine receptor | Muscarinic acetylcholine receptor M2 and M4 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 53079.31 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Cholinergic, muscarinic M4 CHRM4 HUMAN M3::P08173 |
|---|
| Residue: | 479 |
|---|
| Sequence: | MANFTPVNGSSGNQSVRLVTSSSHNRYETVEMVFIATVTGSLSLVTVVGNILVMLSIKVN
RQLQTVNNYFLFSLACADLIIGAFSMNLYTVYIIKGYWPLGAVVCDLWLALDYVVSNASV
MNLLIISFDRYFCVTKPLTYPARRTTKMAGLMIAAAWVLSFVLWAPAILFWQFVVGKRTV
PDNQCFIQFLSNPAVTFGTAIAAFYLPVVIMTVLYIHISLASRSRVHKHRPEGPKEKKAK
TLAFLKSPLMKQSVKKPPPGEAAREELRNGKLEEAPPPALPPPPRPVADKDTSNESSSGS
ATQNTKERPATELSTTEATTPAMPAPPLQPRALNPASRWSKIQIVTKQTGNECVTAIEIV
PATPAGMRPAANVARKFASIARNQVRKKRQMAARERKVTRTIFAILLAFILTWTPYNVMV
LVNTFCQSCIPDTVWSIGYWLCYVNSTINPACYALCNATFKKTFRHLLLCQYRNIGTAR
|
|
|
|---|
| BDBM50048803 |
|---|
| n/a |
|---|
| Name | BDBM50048803 |
|---|
| Synonyms: | 5-(2-(4-(benzo[d]isothiazol-3-yl)piperazin-1-yl)ethyl)-6-chloroindolin-2-one | 5-[2-(4-Benzo[d]isothiazol-3-yl-piperazin-1-yl)-ethyl]-6-chloro-1,3-dihydro-indol-2-one | 5-[2-(4-Benzo[d]isothiazol-3-yl-piperazin-1-yl)-ethyl]-6-chloro-1,3-dihydro-indol-2-one (Ziprasidone) | 5-[2-(4-Benzo[d]isothiazol-3-yl-piperazin-1-yl)-ethyl]-6-chloro-1,3-dihydro-indol-2-one(Norastemizole) | CHEMBL708 | GEODON | ZIPRASIDONE | ZIPRASIDONE HYDROCHLORIDE |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H21ClN4OS |
|---|
| Mol. Mass. | 412.936 |
|---|
| SMILES | Clc1cc2NC(=O)Cc2cc1CCN1CCN(CC1)c1nsc2ccccc12 |
|---|
| Structure | 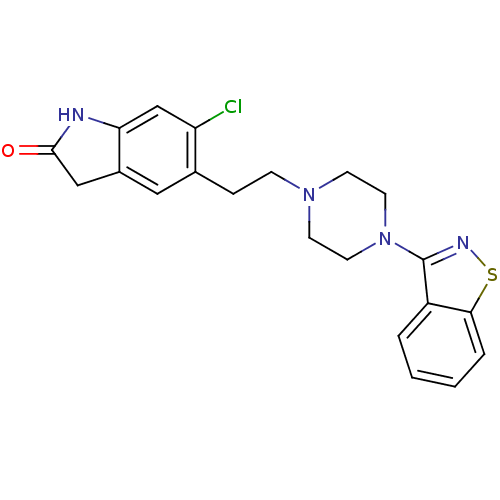
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bymaster, FP; Felder, CC; Tzavara, E; Nomikos, GG; Calligaro, DO; Mckinzie, DL Muscarinic mechanisms of antipsychotic atypicality. Prog Neuropsychopharmacol Biol Psychiatry27:1125-43 (2003) [PubMed] Article
Bymaster, FP; Felder, CC; Tzavara, E; Nomikos, GG; Calligaro, DO; Mckinzie, DL Muscarinic mechanisms of antipsychotic atypicality. Prog Neuropsychopharmacol Biol Psychiatry27:1125-43 (2003) [PubMed] Article