| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein phosphatase non-receptor type 5 |
|---|
| Ligand | BDBM88730 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Dose response confirmation of uHTS small molecule inhibitors of Striatal-Enriched Phosphatase via a fluorescence intensity assay |
|---|
| IC50 | 6730±n/a nM |
|---|
| Citation |  PubChem, PC Dose response confirmation of uHTS small molecule inhibitors of Striatal-Enriched Phosphatase via a fluorescence intensity assay PubChem Bioassay(2012)[AID] PubChem, PC Dose response confirmation of uHTS small molecule inhibitors of Striatal-Enriched Phosphatase via a fluorescence intensity assay PubChem Bioassay(2012)[AID] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein phosphatase non-receptor type 5 |
|---|
| Name: | Tyrosine-protein phosphatase non-receptor type 5 |
|---|
| Synonyms: | PTN5_HUMAN | PTPN5 | Tyrosine-protein phosphatase non-receptor type 5 | Tyrosine-protein phosphatase non-receptor type 5 isoform a |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 63510.68 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | gi_90652859 |
|---|
| Residue: | 565 |
|---|
| Sequence: | MNYEGARSERENHAADDSEGGALDMCCSERLPGLPQPIVMEALDEAEGLQDSQREMPPPP
PPSPPSDPAQKPPPRGAGSHSLTVRSSLCLFAASQFLLACGVLWFSGYGHIWSQNATNLV
SSLLTLLKQLEPTAWLDSGTWGVPSLLLVFLSVGLVLVTTLVWHLLRTPPEPPTPLPPED
RRQSVSRQPSFTYSEWMEEKIEDDFLDLDPVPETPVFDCVMDIKPEADPTSLTVKSMGLQ
ERRGSNVSLTLDMCTPGCNEEGFGYLMSPREESAREYLLSASRVLQAEELHEKALDPFLL
QAEFFEIPMNFVDPKEYDIPGLVRKNRYKTILPNPHSRVCLTSPDPDDPLSSYINANYIR
GYGGEEKVYIATQGPIVSTVADFWRMVWQEHTPIIVMITNIEEMNEKCTEYWPEEQVAYD
GVEITVQKVIHTEDYRLRLISLKSGTEERGLKHYWFTSWPDQKTPDRAPPLLHLVREVEE
AAQQEGPHCAPIIVHCSAGIGRTGCFIATSICCQQLRQEGVVDILKTTCQLRQDRGGMIQ
TCEQYQFVHHVMSLYEKQLSHQSPE
|
|
|
|---|
| BDBM88730 |
|---|
| n/a |
|---|
| Name | BDBM88730 |
|---|
| Synonyms: | MLS000574482 | N-[5-(1H-benzimidazol-2-yl)-2-chloranyl-phenyl]-4-ethoxy-3-nitro-benzamide | N-[5-(1H-benzimidazol-2-yl)-2-chloro-phenyl]-4-ethoxy-3-nitro-benzamide | N-[5-(1H-benzimidazol-2-yl)-2-chlorophenyl]-4-ethoxy-3-nitrobenzamide | SMR000195146 | cid_1338419 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H17ClN4O4 |
|---|
| Mol. Mass. | 436.848 |
|---|
| SMILES | CCOc1ccc(cc1[N+]([O-])=O)C(=O)Nc1cc(ccc1Cl)-c1nc2ccccc2[nH]1 |
|---|
| Structure | 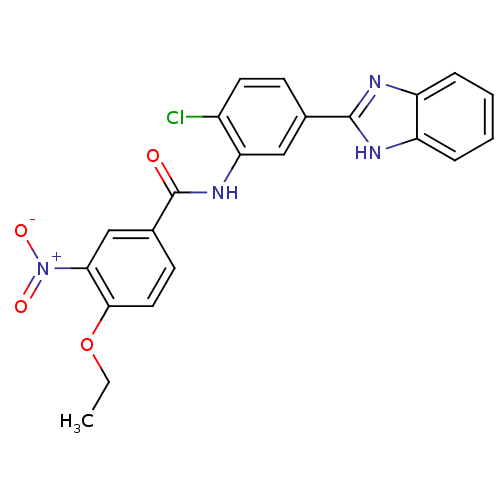
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC Dose response confirmation of uHTS small molecule inhibitors of Striatal-Enriched Phosphatase via a fluorescence intensity assay PubChem Bioassay(2012)[AID]
PubChem, PC Dose response confirmation of uHTS small molecule inhibitors of Striatal-Enriched Phosphatase via a fluorescence intensity assay PubChem Bioassay(2012)[AID]