| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Fatty-acid amide hydrolase 1 |
|---|
| Ligand | BDBM26732 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Inhibition Assay |
|---|
| pH | 8±0 |
|---|
| Temperature | 295.15±0 K |
|---|
| IC50 | 2.3±0.4 nM |
|---|
| Citation |  Tian, G; Paschetto, KA; Gharahdaghi, F; Gordon, E; Wilkins, DE; Luo, X; Scott, CW Mechanism of inhibition of fatty acid amide hydrolase by sulfonamide-containing benzothiazoles: long residence time derived from increased kinetic barrier and not exclusively from thermodynamic potency. Biochemistry50:6867-78 (2011) [PubMed] Article Tian, G; Paschetto, KA; Gharahdaghi, F; Gordon, E; Wilkins, DE; Luo, X; Scott, CW Mechanism of inhibition of fatty acid amide hydrolase by sulfonamide-containing benzothiazoles: long residence time derived from increased kinetic barrier and not exclusively from thermodynamic potency. Biochemistry50:6867-78 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Fatty-acid amide hydrolase 1 |
|---|
| Name: | Fatty-acid amide hydrolase 1 |
|---|
| Synonyms: | Anandamide amidohydrolase | Anandamide amidohydrolase 1 | FAAH | FAAH1 | FAAH1_HUMAN | Fatty Acid Amide Hydrolase (FAAH) | Fatty-acid amide hydrolase (FAAH) | Fatty-acid amide hydrolase 1 | Oleamide hydrolase 1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 63071.19 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | O00519 |
|---|
| Residue: | 579 |
|---|
| Sequence: | MVQYELWAALPGASGVALACCFVAAAVALRWSGRRTARGAVVRARQRQRAGLENMDRAAQ
RFRLQNPDLDSEALLALPLPQLVQKLHSRELAPEAVLFTYVGKAWEVNKGTNCVTSYLAD
CETQLSQAPRQGLLYGVPVSLKECFTYKGQDSTLGLSLNEGVPAECDSVVVHVLKLQGAV
PFVHTNVPQSMFSYDCSNPLFGQTVNPWKSSKSPGGSSGGEGALIGSGGSPLGLGTDIGG
SIRFPSSFCGICGLKPTGNRLSKSGLKGCVYGQEAVRLSVGPMARDVESLALCLRALLCE
DMFRLDPTVPPLPFREEVYTSSQPLRVGYYETDNYTMPSPAMRRAVLETKQSLEAAGHTL
VPFLPSNIPHALETLSTGGLFSDGGHTFLQNFKGDFVDPCLGDLVSILKLPQWLKGLLAF
LVKPLLPRLSAFLSNMKSRSAGKLWELQHEIEVYRKTVIAQWRALDLDVVLTPMLAPALD
LNAPGRATGAVSYTMLYNCLDFPAGVVPVTTVTAEDEAQMEHYRGYFGDIWDKMLQKGMK
KSVGLPVAVQCVALPWQEELCLRFMREVERLMTPEKQSS
|
|
|
|---|
| BDBM26732 |
|---|
| n/a |
|---|
| Name | BDBM26732 |
|---|
| Synonyms: | Benzothiazole-based analogue, 1 | N-[4-(4-methoxy-1,3-benzothiazol-2-yl)phenyl]-1-(thiophene-2-sulfonyl)piperidine-4-carboxamide | benzothiazole analogue, 16j |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H23N3O4S3 |
|---|
| Mol. Mass. | 513.652 |
|---|
| SMILES | COc1cccc2sc(nc12)-c1ccc(NC(=O)C2CCN(CC2)S(=O)(=O)c2cccs2)cc1 |
|---|
| Structure | 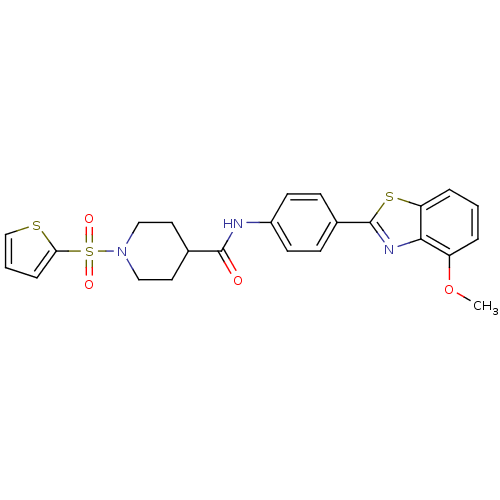
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Tian, G; Paschetto, KA; Gharahdaghi, F; Gordon, E; Wilkins, DE; Luo, X; Scott, CW Mechanism of inhibition of fatty acid amide hydrolase by sulfonamide-containing benzothiazoles: long residence time derived from increased kinetic barrier and not exclusively from thermodynamic potency. Biochemistry50:6867-78 (2011) [PubMed] Article
Tian, G; Paschetto, KA; Gharahdaghi, F; Gordon, E; Wilkins, DE; Luo, X; Scott, CW Mechanism of inhibition of fatty acid amide hydrolase by sulfonamide-containing benzothiazoles: long residence time derived from increased kinetic barrier and not exclusively from thermodynamic potency. Biochemistry50:6867-78 (2011) [PubMed] Article