| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Genome polyprotein |
|---|
| Ligand | BDBM50022180 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | NS5B Inhibition Assay |
|---|
| pH | 7±0 |
|---|
| Temperature | 303.15±0 K |
|---|
| IC50 | 4.72e+4± 5.1e+3 nM |
|---|
| Citation |  Nichols, DB; Leão, RA; Basu, A; Chudayeu, M; de Moraes, Pde F; Talele, TT; Costa, PR; Kaushik-Basu, N Evaluation of Coumarin and Neoflavone Derivatives as HCV NS5B Polymerase Inhibitors. Chem Biol Drug Des81:607-14 (2013) [PubMed] Article Nichols, DB; Leão, RA; Basu, A; Chudayeu, M; de Moraes, Pde F; Talele, TT; Costa, PR; Kaushik-Basu, N Evaluation of Coumarin and Neoflavone Derivatives as HCV NS5B Polymerase Inhibitors. Chem Biol Drug Des81:607-14 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Genome polyprotein |
|---|
| Name: | Genome polyprotein |
|---|
| Synonyms: | Genome polyprotein | Non-structural protein 5B (NS5B) | POLG_HCVBK | RNA-directed RNA polymerase (NS5B) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 327252.46 |
|---|
| Organism: | Hepatitis C virus genotype 1b (isolate BK) (HCV) |
|---|
| Description: | P26663 |
|---|
| Residue: | 3010 |
|---|
| Sequence: | MSTNPKPQRKTKRNTNRRPQDVKFPGGGQIVGGVYLLPRRGPRLGVRAPRKTSERSQPRG
RRQPIPKARRPEGRTWAQPGYPWPLYGNEGLGWAGWLLSPRGSRPSWGPTDPRRRSRNLG
KVIDTLTCGFADLMGYIPLVGAPLGGAARALAHGVRVLEDGVNYATGNLPGCSFSIFLLA
LLSCLTTPASAYEVHNVSGIYHVTNDCSNASIVYEAADLIMHTPGCVPCVREGNSSRCWV
ALTPTLAARNVTIPTTTIRRHVDLLVGAAAFCSAMYVGDLCGSVFLVSQLFTFSPRRHVT
LQDCNCSIYPGHVSGHRMAWDMMMNWSPTTALVVSQLLRIPQAVVDMVAGAHWGVLAGLA
YYSMAGNWAKVLIVMLLFAGVDGDTHVTGGAQAKTTNRLVSMFASGPSQKIQLINTNGSW
HINRTALNCNDSLQTGFLAALFYTHSFNSSGCPERMAQCRTIDKFDQGWGPITYAESSRS
DQRPYCWHYPPPQCTIVPASEVCGPVYCFTPSPVVVGTTDRFGVPTYRWGENETDVLLLN
NTRPPQGNWFGCTWMNSTGFTKTCGGPPCNIGGVGNNTLTCPTDCFRKHPEATYTKCGSG
PWLTPRCMVDYPYRLWHYPCTVNFTIFKVRMYVGGVEHRLNAACNWTRGERCDLEDRDRP
ELSPLLLSTTEWQVLPCSFTTLPALSTGLIHLHQNIVDVQYLYGIGSAVVSFAIKWEYVL
LLFLLLADARVCACLWMMLLIAQAEAALENLVVLNSASVAGAHGILSFLVFFCAAWYIKG
RLVPGATYALYGVWPLLLLLLALPPRAYAMDREMAASCGGAVFVGLVLLTLSPYYKVFLA
RLIWWLQYFTTRAEADLHVWIPPLNARGGRDAIILLMCAVHPELIFDITKLLIAILGPLM
VLQAGITRVPYFVRAQGLIHACMLVRKVAGGHYVQMAFMKLGALTGTYIYNHLTPLRDWP
RAGLRDLAVAVEPVVFSDMETKIITWGADTAACGDIILGLPVSARRGKEILLGPADSLEG
RGLRLLAPITAYSQQTRGLLGCIITSLTGRDKNQVEGEVQVVSTATQSFLATCVNGVCWT
VYHGAGSKTLAAPKGPITQMYTNVDQDLVGWPKPPGARSLTPCTCGSSDLYLVTRHADVI
PVRRRGDSRGSLLSPRPVSYLKGSSGGPLLCPFGHAVGIFRAAVCTRGVAKAVDFVPVES
METTMRSPVFTDNSSPPAVPQSFQVAHLHAPTGSGKSTKVPAAYAAQGYKVLVLNPSVAA
TLGFGAYMSKAHGIDPNIRTGVRTITTGAPVTYSTYGKFLADGGCSGGAYDIIICDECHS
TDSTTILGIGTVLDQAETAGARLVVLATATPPGSVTVPHPNIEEVALSNTGEIPFYGKAI
PIEAIRGGRHLIFCHSKKKCDELAAKLSGLGINAVAYYRGLDVSVIPTIGDVVVVATDAL
MTGYTGDFDSVIDCNTCVTQTVDFSLDPTFTIETTTVPQDAVSRSQRRGRTGRGRRGIYR
FVTPGERPSGMFDSSVLCECYDAGCAWYELTPAETSVRLRAYLNTPGLPVCQDHLEFWES
VFTGLTHIDAHFLSQTKQAGDNFPYLVAYQATVCARAQAPPPSWDQMWKCLIRLKPTLHG
PTPLLYRLGAVQNEVTLTHPITKYIMACMSADLEVVTSTWVLVGGVLAALAAYCLTTGSV
VIVGRIILSGRPAIVPDRELLYQEFDEMEECASHLPYIEQGMQLAEQFKQKALGLLQTAT
KQAEAAAPVVESKWRALETFWAKHMWNFISGIQYLAGLSTLPGNPAIASLMAFTASITSP
LTTQSTLLFNILGGWVAAQLAPPSAASAFVGAGIAGAAVGSIGLGKVLVDILAGYGAGVA
GALVAFKVMSGEMPSTEDLVNLLPAILSPGALVVGVVCAAILRRHVGPGEGAVQWMNRLI
AFASRGNHVSPTHYVPESDAAARVTQILSSLTITQLLKRLHQWINEDCSTPCSGSWLRDV
WDWICTVLTDFKTWLQSKLLPQLPGVPFFSCQRGYKGVWRGDGIMQTTCPCGAQITGHVK
NGSMRIVGPKTCSNTWHGTFPINAYTTGPCTPSPAPNYSRALWRVAAEEYVEVTRVGDFH
YVTGMTTDNVKCPCQVPAPEFFSEVDGVRLHRYAPACRPLLREEVTFQVGLNQYLVGSQL
PCEPEPDVAVLTSMLTDPSHITAETAKRRLARGSPPSLASSSASQLSAPSLKATCTTHHV
SPDADLIEANLLWRQEMGGNITRVESENKVVVLDSFDPLRAEEDEREVSVPAEILRKSKK
FPAAMPIWARPDYNPPLLESWKDPDYVPPVVHGCPLPPIKAPPIPPPRRKRTVVLTESSV
SSALAELATKTFGSSESSAVDSGTATALPDQASDDGDKGSDVESYSSMPPLEGEPGDPDL
SDGSWSTVSEEASEDVVCCSMSYTWTGALITPCAAEESKLPINALSNSLLRHHNMVYATT
SRSAGLRQKKVTFDRLQVLDDHYRDVLKEMKAKASTVKAKLLSVEEACKLTPPHSAKSKF
GYGAKDVRNLSSKAVNHIHSVWKDLLEDTVTPIDTTIMAKNEVFCVQPEKGGRKPARLIV
FPDLGVRVCEKMALYDVVSTLPQVVMGSSYGFQYSPGQRVEFLVNTWKSKKNPMGFSYDT
RCFDSTVTENDIRVEESIYQCCDLAPEARQAIKSLTERLYIGGPLTNSKGQNCGYRRCRA
SGVLTTSCGNTLTCYLKASAACRAAKLQDCTMLVNGDDLVVICESAGTQEDAASLRVFTE
AMTRYSAPPGDPPQPEYDLELITSCSSNVSVAHDASGKRVYYLTRDPTTPLARAAWETAR
HTPVNSWLGNIIMYAPTLWARMILMTHFFSILLAQEQLEKALDCQIYGACYSIEPLDLPQ
IIERLHGLSAFSLHSYSPGEINRVASCLRKLGVPPLRVWRHRARSVRARLLSQGGRAATC
GKYLFNWAVKTKLKLTPIPAASRLDLSGWFVAGYSGGDIYHSLSRARPRWFMLCLLLLSV
GVGIYLLPNR
|
|
|
|---|
| BDBM50022180 |
|---|
| n/a |
|---|
| Name | BDBM50022180 |
|---|
| Synonyms: | 5,7-Dihydroxy-4-methyl-2H-chromen-2-one (2) | 5,7-Dihydroxy-4-methyl-chromen-2-one | 5,7-dihydroxy-4-methyl-2H-chromen-2-one | CHEMBL12252 | Coumarin derivative, 1b |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C10H8O4 |
|---|
| Mol. Mass. | 192.1681 |
|---|
| SMILES | Cc1cc(=O)oc2cc(O)cc(O)c12 |
|---|
| Structure | 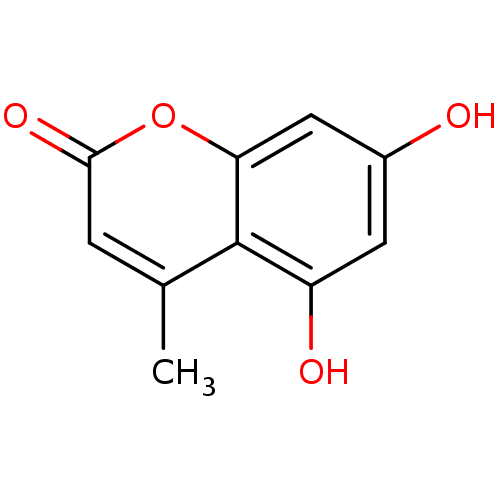
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Nichols, DB; Leão, RA; Basu, A; Chudayeu, M; de Moraes, Pde F; Talele, TT; Costa, PR; Kaushik-Basu, N Evaluation of Coumarin and Neoflavone Derivatives as HCV NS5B Polymerase Inhibitors. Chem Biol Drug Des81:607-14 (2013) [PubMed] Article
Nichols, DB; Leão, RA; Basu, A; Chudayeu, M; de Moraes, Pde F; Talele, TT; Costa, PR; Kaushik-Basu, N Evaluation of Coumarin and Neoflavone Derivatives as HCV NS5B Polymerase Inhibitors. Chem Biol Drug Des81:607-14 (2013) [PubMed] Article