| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Xylosyl- and glucuronyltransferase LARGE1 |
|---|
| Ligand | BDBM64392 |
|---|
| Substrate/Competitor | n/a |
|---|
| EC50 | 8890±n/a nM |
|---|
| Citation |  PubChem, PC Dose response confirmation of small molecule activators of alpha dystroglycan glycosylation PubChem Bioassay(2012)[AID] PubChem, PC Dose response confirmation of small molecule activators of alpha dystroglycan glycosylation PubChem Bioassay(2012)[AID] |
|---|
| More Info.: | Get all data from this article |
|---|
| |
| Xylosyl- and glucuronyltransferase LARGE1 |
|---|
| Name: | Xylosyl- and glucuronyltransferase LARGE1 |
|---|
| Synonyms: | KIAA0609 | LARG1_HUMAN | LARGE | LARGE1 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 88079.63 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | gi_47678551 |
|---|
| Residue: | 756 |
|---|
| Sequence: | MLGICRGRRKFLAASLSLLCIPAITWIYLFSGSFEDGKPVSLSPLESQAHSPRYTASSQR
ERESLEVRMREVEEENRALRRQLSLAQGRAPSHRRGNHSKTYSMEEGTGDSENLRAGIVA
GNSSECGQQPVVEKCETIHVAIVCAGYNASRDVVTLVKSVLFHRRNPLHFHLIADSIAEQ
ILATLFQTWMVPAVRVDFYNADELKSEVSWIPNKHYSGIYGLMKLVLTKTLPANLERVIV
LDTDITFATDIAELWAVFHKFKGQQVLGLVENQSDWYLGNLWKNHRPWPALGRGYNTGVI
LLLLDKLRKMKWEQMWRLTAERELMGMLSTSLADQDIFNAVIKQNPFLVYQLPCFWNVQL
SDHTRSEQCYRDVSDLKVIHWNSPKKLRVKNKHVEFFRNLYLTFLEYDGNLLRRELFGCP
SEADVNSENLQKQLSELDEDDLCYEFRRERFTVHRTHLYFLHYEYEPAADSTDVTLVAQL
SMDRLQMLEAICKHWEGPISLALYLSDAEAQQFLRYAQGSEVLMSRHNVGYHIVYKEGQF
YPVNLLRNVAMKHISTPYMFLSDIDFLPMYGLYEYLRKSVIQLDLANTKKAMIVPAFETL
RYRLSFPKSKAELLSMLDMGTLFTFRYHVWTKGHAPTNFAKWRTATTPYRVEWEADFEPY
VVVRRDCPEYDRRFVGFGWNKVAHIMELDVQEYEFIVLPNAYMIHMPHAPSFDITKFRSN
KQYRICLKTLKEEFQQDMSRRYGFAALKYLTAENNS
|
|
|
|---|
| BDBM64392 |
|---|
| n/a |
|---|
| Name | BDBM64392 |
|---|
| Synonyms: | 1-(4-methoxy-2,3-dimethyl-benzyl)-N-[4-(3-pyridyloxy)phenyl]isonipecotamide | 1-[(4-methoxy-2,3-dimethyl-phenyl)methyl]-N-(4-pyridin-3-yloxyphenyl)piperidine-4-carboxamide | 1-[(4-methoxy-2,3-dimethylphenyl)methyl]-N-(4-pyridin-3-yloxyphenyl)piperidine-4-carboxamide | 1-[(4-methoxy-2,3-dimethylphenyl)methyl]-N-[4-(3-pyridinyloxy)phenyl]-4-piperidinecarboxamide | MLS001090782 | SMR000653599 | cid_24817212 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H31N3O3 |
|---|
| Mol. Mass. | 445.5533 |
|---|
| SMILES | COc1ccc(CN2CCC(CC2)C(=O)Nc2ccc(Oc3cccnc3)cc2)c(C)c1C |
|---|
| Structure | 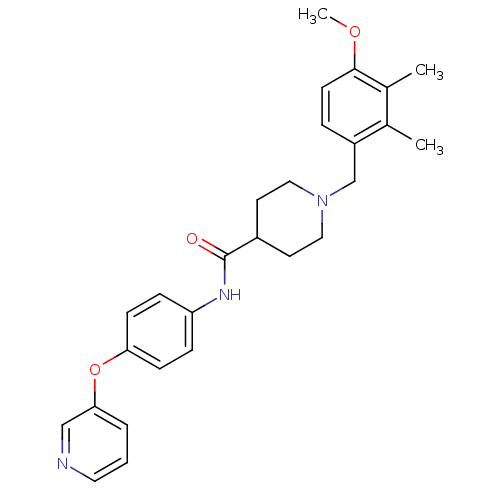
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC Dose response confirmation of small molecule activators of alpha dystroglycan glycosylation PubChem Bioassay(2012)[AID]
PubChem, PC Dose response confirmation of small molecule activators of alpha dystroglycan glycosylation PubChem Bioassay(2012)[AID]