| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50015234 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | In vitro CYP3A4 Inhibition Assay |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Bareja, SP; Jones, SD; Oblong, JE; Mills, KJ; Bierman, JC Cosmetic compositions containing substituted azole and methods for alleviating the signs of photoaged skin US Patent US9144538 Publication Date 9/29/2015 Bareja, SP; Jones, SD; Oblong, JE; Mills, KJ; Bierman, JC Cosmetic compositions containing substituted azole and methods for alleviating the signs of photoaged skin US Patent US9144538 Publication Date 9/29/2015 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50015234 |
|---|
| n/a |
|---|
| Name | BDBM50015234 |
|---|
| Synonyms: | 4,4''[1,6-HEXANEDIYLBIS(OXY)]BISBENZENECARBOXIMIDAMIDE | 4-[6-(4-Carbamimidoyl-phenoxy)-hexyloxy]-benzamidine | 4-{6-[4-amino(imino)methylphenoxy]hexyloxy}phenyl-iminomethanamine | 4-{6-[4-ammino(imino)methylphenoxy]hexyloxy}phenyl-iminomethanammine | CHEMBL25105 | Hexamidine | US9138393, Hexamidine diisethionate | US9144538, Hexamidine diisethionate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H26N4O2 |
|---|
| Mol. Mass. | 354.446 |
|---|
| SMILES | NC(=N)c1ccc(OCCCCCCOc2ccc(cc2)C(N)=N)cc1 |
|---|
| Structure | 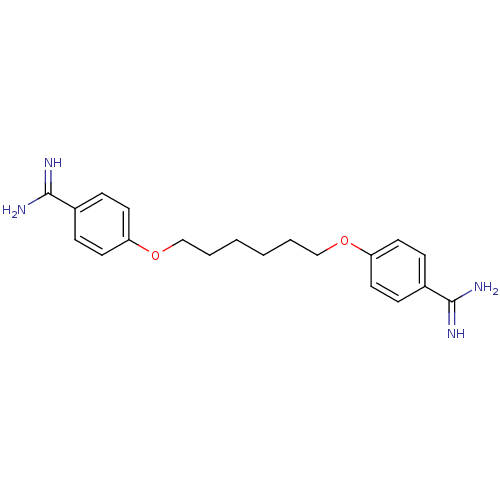
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bareja, SP; Jones, SD; Oblong, JE; Mills, KJ; Bierman, JC Cosmetic compositions containing substituted azole and methods for alleviating the signs of photoaged skin US Patent US9144538 Publication Date 9/29/2015
Bareja, SP; Jones, SD; Oblong, JE; Mills, KJ; Bierman, JC Cosmetic compositions containing substituted azole and methods for alleviating the signs of photoaged skin US Patent US9144538 Publication Date 9/29/2015