| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Nitric oxide synthase, endothelial |
|---|
| Ligand | BDBM50328814 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Inhibition Assay |
|---|
| pH | 7.4±n/a |
|---|
| Ki | 19200±n/a nM |
|---|
| Comments | extracted |
|---|
| Citation |  Silverman, RB; Cinelli, MA 2-aminoquinoline-based compounds for potent and selective neuronal nitric oxide synthase inhibition US Patent US9212144 Publication Date 12/15/2015 Silverman, RB; Cinelli, MA 2-aminoquinoline-based compounds for potent and selective neuronal nitric oxide synthase inhibition US Patent US9212144 Publication Date 12/15/2015 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Nitric oxide synthase, endothelial |
|---|
| Name: | Nitric oxide synthase, endothelial |
|---|
| Synonyms: | Constitutive NOS | Endothelial NOS | Endothelial nitric oxide synthase | Endothelial nitric-oxide synthase (eNOS) | NOS type III | NOS3 | NOS3_BOVIN | Nitric oxide synthase, endothelial (eNOS) | Nitric-oxide synthase, endothelial | cNOS |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 133292.26 |
|---|
| Organism: | Bos taurus (bovine) |
|---|
| Description: | Recombinant eNOS overexpressed in E. coli was used in enzyme assays. |
|---|
| Residue: | 1205 |
|---|
| Sequence: | MGNLKSVGQEPGPPCGLGLGLGLGLCGKQGPASPAPEPSRAPAPATPHAPDHSPAPNSPT
LTRPPEGPKFPRVKNWELGSITYDTLCAQSQQDGPCTPRCCLGSLVLPRKLQTRPSPGPP
PAEQLLSQARDFINQYYSSIKRSGSQAHEERLQEVEAEVASTGTYHLRESELVFGAKQAW
RNAPRCVGRIQWGKLQVFDARDCSSAQEMFTYICNHIKYATNRGNLRSAITVFPQRAPGR
GDFRIWNSQLVRYAGYRQQDGSVRGDPANVEITELCIQHGWTPGNGRFDVLPLLLQAPDE
APELFVLPPELVLEVPLEHPTLEWFAALGLRWYALPAVSNMLLEIGGLEFSAAPFSGWYM
STEIGTRNLCDPHRYNILEDVAVCMDLDTRTTSSLWKDKAAVEINLAVLHSFQLAKVTIV
DHHAATVSFMKHLDNEQKARGGCPADWAWIVPPISGSLTPVFHQEMVNYILSPAFRYQPD
PWKGSATKGAGITRKKTFKEVANAVKISASLMGTLMAKRVKATILYASETGRAQSYAQQL
GRLFRKAFDPRVLCMDEYDVVSLEHEALVLVVTSTFGNGDPPENGESFAAALMEMSGPYN
SSPRPEQHKSYKIRFNSVSCSDPLVSSWRRKRKESSNTDSAGALGTLRFCVFGLGSRAYP
HFCAFARAVDTRLEELGGERLLQLGQGDELCGQEEAFRGWAKAAFQASCETFCVGEEAKA
AAQDIFSPKRSWKRQRYRLSTQAEGLQLLPGLIHVHRRKMFQATVLSVENLQSSKSTRAT
ILVRLDTAGQEGLQYQPGDHIGICPPNRPGLVEALLSRVEDPPPPTESVAVEQLEKGSPG
GPPPSWVRDPRLPPCTLRQALTFFLDITSPPSPRLLRLLSTLAEEPSEQQELETLSQDPR
RYEEWKWFRCPTLLEVLEQFPSVALPAPLLLTQLPLLQPRYYSVSSAPNAHPGEVHLTVA
VLAYRTQDGLGPLHYGVCSTWLSQLKTGDPVPCFIRGAPSFRLPPDPYVPCILVGPGTGI
APFRGFWQERLHDIESKGLQPAPMTLVFGCRCSQLDHLYRDEVQDAQERGVFGRVLTAFS
REPDSPKTYVQDILRTELAAEVHRVLCLERGHMFVCGDVTMATSVLQTVQRILATEGDME
LDEAGDVIGVLRDQQRYHEDIFGLTLRTQEVTSRIRTQSFSLQERHLRGAVPWAFDPPGP
DTPGP
|
|
|
|---|
| BDBM50328814 |
|---|
| n/a |
|---|
| Name | BDBM50328814 |
|---|
| Synonyms: | (+/-)-6-((4-(2-(3-fluorophenethylamino)ethoxy)pyrrolidin-3-yl)methyl)-4-methylpyridin-2-amine | 6-(((3R,4R)-4-(2-(3-fluorophenethylamino)ethoxy)pyrrolidin-3-yl)methyl)-4-methylpyridin-2-amine | 6-{[(3R,4R)-4-(2-{[2-(3-fluorophenyl)ethyl]amino}ethoxy)pyrrolidin-3-yl]methyl}-4-methylpyridin-2-amine | 6-{{(3''R,4''R)-3''-[2''''-(3''''''-Fluorophenethylamino)ethoxy]pyrrolidin-4''-yl}methyl}-4-methylpyridin-2-amine Trihydrochloride | CHEMBL1278132 | CHEMBL607146 | US9212144, 2 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H29FN4O |
|---|
| Mol. Mass. | 372.4796 |
|---|
| SMILES | Cc1cc(N)nc(C[C@@H]2CNC[C@@H]2OCCNCCc2cccc(F)c2)c1 |r| |
|---|
| Structure | 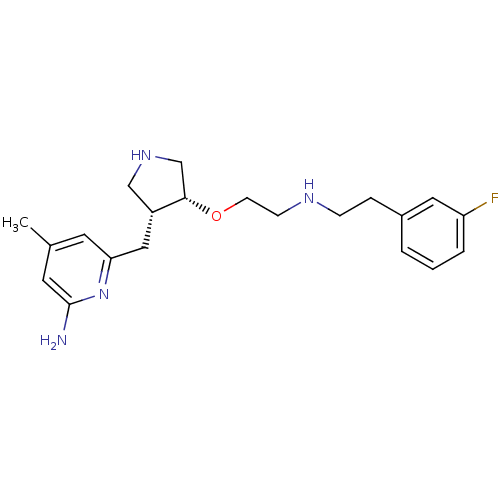
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Silverman, RB; Cinelli, MA 2-aminoquinoline-based compounds for potent and selective neuronal nitric oxide synthase inhibition US Patent US9212144 Publication Date 12/15/2015
Silverman, RB; Cinelli, MA 2-aminoquinoline-based compounds for potent and selective neuronal nitric oxide synthase inhibition US Patent US9212144 Publication Date 12/15/2015