

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Ghrelin O-acyltransferase | ||
| Ligand | BDBM80086 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | GOAT Activity Assay | ||
| pH | 7±n/a | ||
| Temperature | 298.15±n/a K | ||
| IC50 | 3.8e+4± 6e+3 nM | ||
| Comments | extracted | ||
| Citation |  McGovern-Gooch, KR; Mahajani, NS; Garagozzo, A; Schramm, AJ; Hannah, LG; Sieburg, MA; Chisholm, JD; Hougland, JL Synthetic Triterpenoid Inhibition of Human Ghrelin O-Acyltransferase: The Involvement of a Functionally Required Cysteine Provides Mechanistic Insight into Ghrelin Acylation. Biochemistry56:919-931 (2017) [PubMed] Article McGovern-Gooch, KR; Mahajani, NS; Garagozzo, A; Schramm, AJ; Hannah, LG; Sieburg, MA; Chisholm, JD; Hougland, JL Synthetic Triterpenoid Inhibition of Human Ghrelin O-Acyltransferase: The Involvement of a Functionally Required Cysteine Provides Mechanistic Insight into Ghrelin Acylation. Biochemistry56:919-931 (2017) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Ghrelin O-acyltransferase | |||
| Name: | Ghrelin O-acyltransferase | ||
| Synonyms: | GOAT | Ghrelin O-acyltransferase (GOAT) | MBOA4_HUMAN | MBOAT4 | Membrane-bound O-acyltransferase domain-containing protein 4 | O-acyltransferase domain-containing protein 4 | OACT4 | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 49733.10 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | Q96T53 | ||
| Residue: | 435 | ||
| Sequence: |
| ||
| BDBM80086 | |||
| n/a | |||
| Name | BDBM80086 | ||
| Synonyms: | (4aR,6aR,6aS,6bR,8aS,12aS,14bS)-8a-(imidazole-1-carbonyl)-3,13-diketo-4,4,6a,6b,11,11,14b-heptamethyl-4a,5,6,6a,7,8,9,10,12,12a-decahydropicene-2-carbonitrile | (4aR,6aR,6aS,6bR,8aS,12aS,14bS)-8a-(imidazole-1-carbonyl)-4,4,6a,6b,11,11,14b-heptamethyl-3,13-dioxo-4a,5,6,6a,7,8,9,10,12,12a-decahydropicene-2-carbonitrile | (4aR,6aR,6aS,6bR,8aS,12aS,14bS)-8a-[1-imidazolyl(oxo)methyl]-4,4,6a,6b,11,11,14b-heptamethyl-3,13-dioxo-4a,5,6,6a,7,8,9,10,12,12a-decahydropicene-2-carbonitrile | (4aR,6aR,6aS,6bR,8aS,12aS,14bS)-8a-imidazol-1-ylcarbonyl-4,4,6a,6b,11,11,14b-heptamethyl-3,13-bis(oxidanylidene)-4a,5,6,6a,7,8,9,10,12,12a-decahydropicene-2-carbonitrile | CDDO-Im, 1 | MLS002702501 | SMR001566062 | cid_9958995 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C34H43N3O3 | ||
| Mol. Mass. | 541.7235 | ||
| SMILES | CC1(C)CC[C@@]2(CC[C@]3(C)[C@@H]([C@@H]2C1)C(=O)C=C1[C@@]2(C)C=C(C#N)C(=O)C(C)(C)[C@@H]2CC[C@@]31C)C(=O)n1ccnc1 |t:17,21| | ||
| Structure | 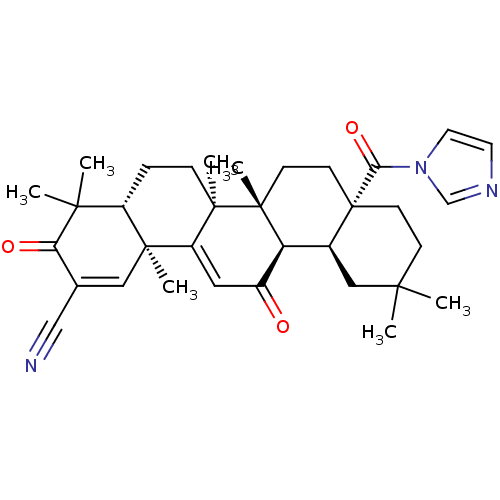
| ||