| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Beta-glucuronidase |
|---|
| Ligand | BDBM163642 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Cell-Based Assay |
|---|
| IC50 | 1688.9±24.12 nM |
|---|
| Citation |  Redinbo, MR Selective β-glucuronidase inhibitors as a treatment for side effects of camptothecin antineoplastic agents US Patent US9334288 Publication Date 5/10/2016 Redinbo, MR Selective β-glucuronidase inhibitors as a treatment for side effects of camptothecin antineoplastic agents US Patent US9334288 Publication Date 5/10/2016 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Beta-glucuronidase |
|---|
| Name: | Beta-glucuronidase |
|---|
| Synonyms: | β-Glucuronidase | BGLR_HUMAN | Beta-G1 | Beta-glucuronidase | GUSB | beta-Glucuronidase (β-glucuronidase) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 74736.05 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 651 |
|---|
| Sequence: | MARGSAVAWAALGPLLWGCALGLQGGMLYPQESPSRECKELDGLWSFRADFSDNRRRGFE
EQWYRRPLWESGPTVDMPVPSSFNDISQDWRLRHFVGWVWYEREVILPERWTQDLRTRVV
LRIGSAHSYAIVWVNGVDTLEHEGGYLPFEADISNLVQVGPLPSRLRITIAINNTLTPTT
LPPGTIQYLTDTSKYPKGYFVQNTYFDFFNYAGLQRSVLLYTTPTTYIDDITVTTSVEQD
SGLVNYQISVKGSNLFKLEVRLLDAENKVVANGTGTQGQLKVPGVSLWWPYLMHERPAYL
YSLEVQLTAQTSLGPVSDFYTLPVGIRTVAVTKSQFLINGKPFYFHGVNKHEDADIRGKG
FDWPLLVKDFNLLRWLGANAFRTSHYPYAEEVMQMCDRYGIVVIDECPGVGLALPQFFNN
VSLHHHMQVMEEVVRRDKNHPAVVMWSVANEPASHLESAGYYLKMVIAHTKSLDPSRPVT
FVSNSNYAADKGAPYVDVICLNSYYSWYHDYGHLELIQLQLATQFENWYKKYQKPIIQSE
YGAETIAGFHQDPPLMFTEEYQKSLLEQYHLGLDQKRRKYVVGELIWNFADFMTEQSPTR
VLGNKKGIFTRQRQPKSAAFLLRERYWKIANETRYPHSVAKSQCLENSLFT
|
|
|
|---|
| BDBM163642 |
|---|
| n/a |
|---|
| Name | BDBM163642 |
|---|
| Synonyms: | Methyl 2amino5[(3chloro4methylphenyl)carbamoyl]4methylthiophene3carboxylate (Inh 7) | US9334288, 3 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H15ClN2O3S |
|---|
| Mol. Mass. | 338.809 |
|---|
| SMILES | COC(=O)c1c(N)sc(C(=O)Nc2ccc(C)c(Cl)c2)c1C |
|---|
| Structure | 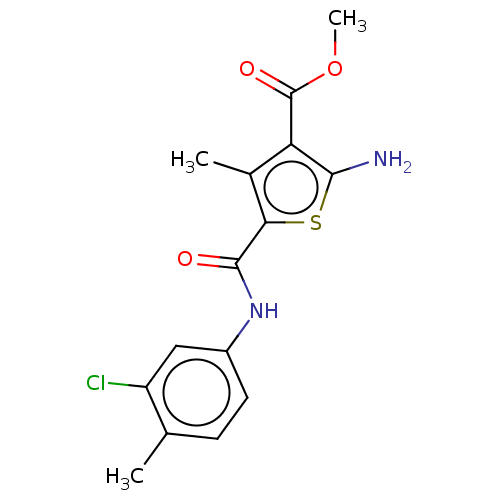
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Redinbo, MR Selective β-glucuronidase inhibitors as a treatment for side effects of camptothecin antineoplastic agents US Patent US9334288 Publication Date 5/10/2016
Redinbo, MR Selective β-glucuronidase inhibitors as a treatment for side effects of camptothecin antineoplastic agents US Patent US9334288 Publication Date 5/10/2016