| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Siderophore-binding protein |
|---|
| Ligand | BDBM11622 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | CA Inhibition Assay |
|---|
| pH | 8.3±0 |
|---|
| Temperature | 298.15±0 K |
|---|
| Ki | 1.7e+2±n/a nM |
|---|
| Citation |  Maresca, A; Scozzafava, A; Vullo, D; Supuran, CT Dihalogenated sulfanilamides and benzolamides are effective inhibitors of the three ß-class carbonic anhydrases from Mycobacterium tuberculosis. J Enzyme Inhib Med Chem28:384-7 (2013) [PubMed] Article Maresca, A; Scozzafava, A; Vullo, D; Supuran, CT Dihalogenated sulfanilamides and benzolamides are effective inhibitors of the three ß-class carbonic anhydrases from Mycobacterium tuberculosis. J Enzyme Inhib Med Chem28:384-7 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Siderophore-binding protein |
|---|
| Name: | Siderophore-binding protein |
|---|
| Synonyms: | β-Carbonic anhydrase 3 (CA 3) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 17663.22 |
|---|
| Organism: | Mycobacterium tuberculosis |
|---|
| Description: | n/a |
|---|
| Residue: | 174 |
|---|
| Sequence: | MPLFSFEGRSPRIDPTAFVAPTATLIGDVTIEAGASVWFNAVLRGDYAPVVVREGANVQD
GAVLHAPPGIPVDIGPGATVAHLCVIHGVHVGSEALIANHATVLDGAVIGARCMIAAGAL
VVAGTQIPAGMLVTGAPAKVKGPIEGTGAEMWVNVNPQAYRDLAARHLAGLEPM
|
|
|
|---|
| BDBM11622 |
|---|
| n/a |
|---|
| Name | BDBM11622 |
|---|
| Synonyms: | β-CA inhibitor, 1 | 2-N-(4-amino-3-chlorobenzene)-1,3,4-thiadiazole-2,5-disulfonamide | 5-(4-Amino-3-chlorobenzenesulfonamido)-1,3,4-thiadiazole-2-sulfonamide | aminobenzolamide 17c |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C8H8ClN5O4S3 |
|---|
| Mol. Mass. | 369.828 |
|---|
| SMILES | Nc1ccc(cc1Cl)S(=O)(=O)Nc1nnc(s1)S(N)(=O)=O |
|---|
| Structure | 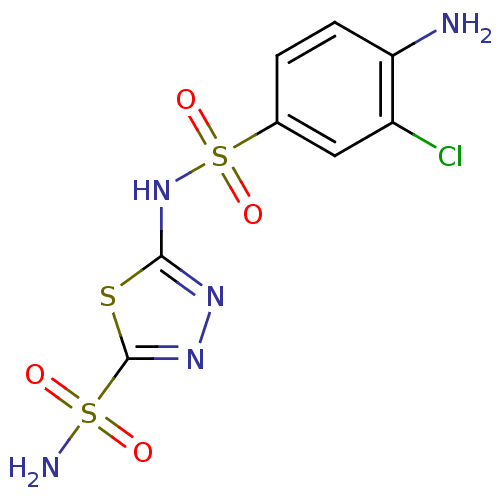
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Maresca, A; Scozzafava, A; Vullo, D; Supuran, CT Dihalogenated sulfanilamides and benzolamides are effective inhibitors of the three ß-class carbonic anhydrases from Mycobacterium tuberculosis. J Enzyme Inhib Med Chem28:384-7 (2013) [PubMed] Article
Maresca, A; Scozzafava, A; Vullo, D; Supuran, CT Dihalogenated sulfanilamides and benzolamides are effective inhibitors of the three ß-class carbonic anhydrases from Mycobacterium tuberculosis. J Enzyme Inhib Med Chem28:384-7 (2013) [PubMed] Article