| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 6 |
|---|
| Ligand | BDBM255474 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Binding Assay or Functional Activity Assay |
|---|
| Ki | >5830±n/a nM |
|---|
| Citation |  Ledgard, AJ (Thieno[2,3-b][1,5]benzoxazepin-4-yl)piperazin-1-yl compounds as dual activity H1 inverse agonists/5-HT2A antagonists US Patent US9481688 Publication Date 11/1/2016 Ledgard, AJ (Thieno[2,3-b][1,5]benzoxazepin-4-yl)piperazin-1-yl compounds as dual activity H1 inverse agonists/5-HT2A antagonists US Patent US9481688 Publication Date 11/1/2016 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 6 |
|---|
| Name: | 5-hydroxytryptamine receptor 6 |
|---|
| Synonyms: | 5-HT-6 | 5-HT6 | 5-hydroxytryptamine receptor 6 (5-HT-6) | 5-hydroxytryptamine receptor 6 (5-HT6R) | 5-hydroxytryptamine receptor 6 (5HT6) | 5HT6R_HUMAN | HTR6 | Serotonin (5-HT3) receptor | Serotonin 6 (5-HT6) receptor | Serotonin Receptor 6 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 46968.43 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P50406 |
|---|
| Residue: | 440 |
|---|
| Sequence: | MVPEPGPTANSTPAWGAGPPSAPGGSGWVAAALCVVIALTAAANSLLIALICTQPALRNT
SNFFLVSLFTSDLMVGLVVMPPAMLNALYGRWVLARGLCLLWTAFDVMCCSASILNLCLI
SLDRYLLILSPLRYKLRMTPLRALALVLGAWSLAALASFLPLLLGWHELGHARPPVPGQC
RLLASLPFVLVASGLTFFLPSGAICFTYCRILLAARKQAVQVASLTTGMASQASETLQVP
RTPRPGVESADSRRLATKHSRKALKASLTLGILLGMFFVTWLPFFVANIVQAVCDCISPG
LFDVLTWLGYCNSTMNPIIYPLFMRDFKRALGRFLPCPRCPRERQASLASPSLRTSHSGP
RPGLSLQQVLPLPLPPDSDSDSDAGSGGSSGLRLTAQLLLPGEATQDPPLPTRAAAAVNF
FNIDPAEPELRPHPLGIPTN
|
|
|
|---|
| BDBM255474 |
|---|
| n/a |
|---|
| Name | BDBM255474 |
|---|
| Synonyms: | US9481688, 1 | US9481688, 3 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H24ClN3O3S |
|---|
| Mol. Mass. | 433.952 |
|---|
| SMILES | Cc1ccc2N=C(N3CCN(CC(C)(C)C(O)=O)CC3)c3cc(Cl)sc3Oc2c1 |t:5| |
|---|
| Structure | 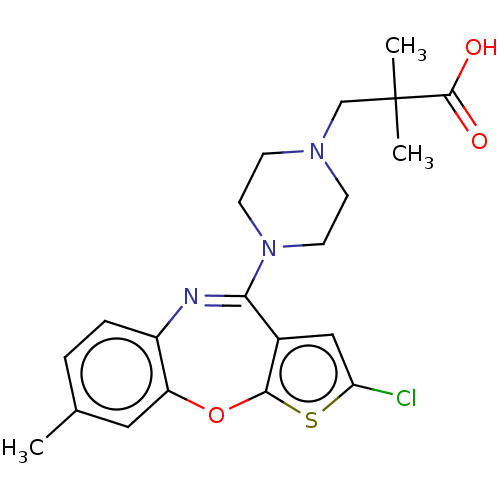
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ledgard, AJ (Thieno[2,3-b][1,5]benzoxazepin-4-yl)piperazin-1-yl compounds as dual activity H1 inverse agonists/5-HT2A antagonists US Patent US9481688 Publication Date 11/1/2016
Ledgard, AJ (Thieno[2,3-b][1,5]benzoxazepin-4-yl)piperazin-1-yl compounds as dual activity H1 inverse agonists/5-HT2A antagonists US Patent US9481688 Publication Date 11/1/2016