| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histone-lysine N-methyltransferase EZH2 [2-746,Y641N] |
|---|
| Ligand | BDBM50246953 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Biological Assay |
|---|
| IC50 | 302±n/a nM |
|---|
| Citation |  Collins, MR; Kania, RS; Kumpf, RA; Kung, P; Richter, DT; Sutton, SC; Wythes, MJ Substituted dihydroisoquinolinone compounds US Patent US10570121 Publication Date 2/25/2020 Collins, MR; Kania, RS; Kumpf, RA; Kung, P; Richter, DT; Sutton, SC; Wythes, MJ Substituted dihydroisoquinolinone compounds US Patent US10570121 Publication Date 2/25/2020 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Histone-lysine N-methyltransferase EZH2 [2-746,Y641N] |
|---|
| Name: | Histone-lysine N-methyltransferase EZH2 [2-746,Y641N] |
|---|
| Synonyms: | EZH2 | EZH2_HUMAN | Histone-lysine N-methyltransferase EZH2 (Y641N) | KMT6 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 85187.58 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q15910 Y641N |
|---|
| Residue: | 745 |
|---|
| Sequence: | GQTGKKSEKGPVCWRKRVKSEYMRLRQLKRFRRADEVKSMFSSNRQKILERTEILNQEWK
QRRIQPVHILTSVSSLRGTRECSVTSDLDFPTQVIPLKTLNAVASVPIMYSWSPLQQNFM
VEDETVLHNIPYMGDEVLDQDGTFIEELIKNYDGKVHGDRECGFINDEIFVELVNALGQY
NDDDDDDDGDDPEEREEKQKDLEDHRDDKESRPPRKFPSDKIFEAISSMFPDKGTAEELK
EKYKELTEQQLPGALPPECTPNIDGPNAKSVQREQSLHSFHTLFCRRCFKYDCFLHPFHA
TPNTYKRKNTETALDNKPCGPQCYQHLEGAKEFAAALTAERIKTPPKRPGGRRRGRLPNN
SSRPSTPTINVLESKDTDSDREAGTETGGENNDKEEEEKKDETSSSSEANSRCQTPIKMK
PNIEPPENVEWSGAEASMFRVLIGTYYDNFCAIARLIGTKTCRQVYEFRVKESSIIAPAP
AEDVDTPPRKKKRKHRLWAAHCRKIQLKKDGSSNHVYNYQPCDHPRQPCDSSCPCVIAQN
FCEKFCQCSSECQNRFPGCRCKAQCNTKQCPCYLAVRECDPDLCLTCGAADHWDSKNVSC
KNCSIQRGSKKHLLLAPSDVAGWGIFIKDPVQKNEFISENCGEIISQDEADRRGKVYDKY
MCSFLFNLNNDFVVDATRKGNKIRFANHSVNPNCYAKVMMVNGDHRIGIFAKRAIQTGEE
LFFDYRYSQADALKYVGIEREMEIP
|
|
|
|---|
| BDBM50246953 |
|---|
| n/a |
|---|
| Name | BDBM50246953 |
|---|
| Synonyms: | CHEMBL4090352 | US10570121, Example 3 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H26Cl2N2O4 |
|---|
| Mol. Mass. | 465.37 |
|---|
| SMILES | Cc1cc(C)c(CN2CCc3c(Cl)cc(C(CO)C4CCOC4)c(Cl)c3C2=O)c(=O)[nH]1 |
|---|
| Structure | 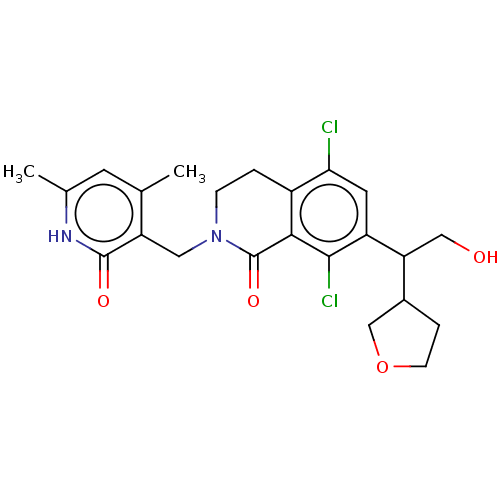
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Collins, MR; Kania, RS; Kumpf, RA; Kung, P; Richter, DT; Sutton, SC; Wythes, MJ Substituted dihydroisoquinolinone compounds US Patent US10570121 Publication Date 2/25/2020
Collins, MR; Kania, RS; Kumpf, RA; Kung, P; Richter, DT; Sutton, SC; Wythes, MJ Substituted dihydroisoquinolinone compounds US Patent US10570121 Publication Date 2/25/2020