| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Interferon-induced, double-stranded RNA-activated protein kinase |
|---|
| Ligand | BDBM365353 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | PKR KinaseGlo Assay |
|---|
| IC50 | 390±n/a nM |
|---|
| Citation |  Boral, S; Malone, TC; Wang, S Pyridyl benzothiophenes as kinase inhibitors US Patent US10669265 Publication Date 6/2/2020 Boral, S; Malone, TC; Wang, S Pyridyl benzothiophenes as kinase inhibitors US Patent US10669265 Publication Date 6/2/2020 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Interferon-induced, double-stranded RNA-activated protein kinase |
|---|
| Name: | Interferon-induced, double-stranded RNA-activated protein kinase |
|---|
| Synonyms: | E2AK2_HUMAN | EIF2AK2 | Eukaryotic translation initiation factor 2-alpha kinase 2 | Interferon-inducible RNA-dependent protein kinase | P1/eIF-2A protein kinase | PKR | PRKR | Protein kinase R | Protein kinase RNA-activated | Tyrosine-protein kinase EIF2AK2 | eIF-2A protein kinase 2 | p68 kinase |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 62104.61 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1467743 |
|---|
| Residue: | 551 |
|---|
| Sequence: | MAGDLSAGFFMEELNTYRQKQGVVLKYQELPNSGPPHDRRFTFQVIIDGREFPEGEGRSK
KEAKNAAAKLAVEILNKEKKAVSPLLLTTTNSSEGLSMGNYIGLINRIAQKKRLTVNYEQ
CASGVHGPEGFHYKCKMGQKEYSIGTGSTKQEAKQLAAKLAYLQILSEETSVKSDYLSSG
SFATTCESQSNSLVTSTLASESSSEGDFSADTSEINSNSDSLNSSSLLMNGLRNNQRKAK
RSLAPRFDLPDMKETKYTVDKRFGMDFKEIELIGSGGFGQVFKAKHRIDGKTYVIKRVKY
NNEKAEREVKALAKLDHVNIVHYNGCWDGFDYDPETSDDSLESSDYDPENSKNSSRSKTK
CLFIQMEFCDKGTLEQWIEKRRGEKLDKVLALELFEQITKGVDYIHSKKLIHRDLKPSNI
FLVDTKQVKIGDFGLVTSLKNDGKRTRSKGTLRYMSPEQISSQDYGKEVDLYALGLILAE
LLHVCDTAFETSKFFTDLRDGIISDIFDKKEKTLLQKLLSKKPEDRPNTSEILRTLTVWK
KSPEKNERHTC
|
|
|
|---|
| BDBM365353 |
|---|
| n/a |
|---|
| Name | BDBM365353 |
|---|
| Synonyms: | 5-{5-[({[4-chloro-3- (trifluoromethyl)phenyl]amino} carbonyl)amino]-1- benzothien-2-yl}-N- [dimethyl(oxido)-lambda4- sulfanylidene]nicotinamide | US10633373, Example 58 | US10669265, Example 58 | US9868726, Example 58 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H18ClF3N4O3S2 |
|---|
| Mol. Mass. | 567.003 |
|---|
| SMILES | CS(C)(=O)=NC(=O)c1cncc(c1)-c1cc2cc(NC(=O)Nc3ccc(Cl)c(c3)C(F)(F)F)ccc2s1 |
|---|
| Structure | 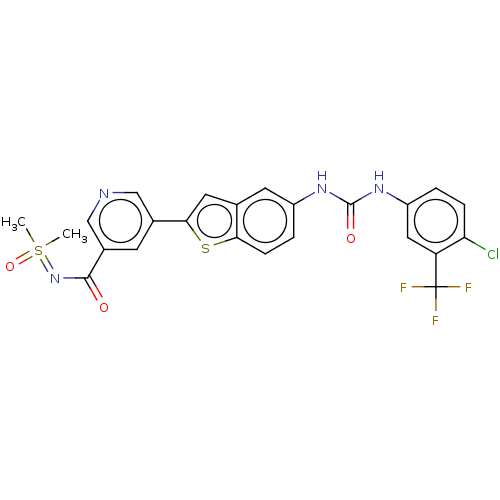
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Boral, S; Malone, TC; Wang, S Pyridyl benzothiophenes as kinase inhibitors US Patent US10669265 Publication Date 6/2/2020
Boral, S; Malone, TC; Wang, S Pyridyl benzothiophenes as kinase inhibitors US Patent US10669265 Publication Date 6/2/2020