| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2A |
|---|
| Ligand | BDBM458718 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Binding Activity Assay |
|---|
| Ki | 0.340±n/a nM |
|---|
| Citation |  Yoshinaga, H; Ikuma, Y; Ikeda, J; Adachi, S; Mitsunuma, H; Aihara, Y; Besnard, J; Bell, AS Condensed lactam derivative US Patent US10745401 Publication Date 8/18/2020 Yoshinaga, H; Ikuma, Y; Ikeda, J; Adachi, S; Mitsunuma, H; Aihara, Y; Besnard, J; Bell, AS Condensed lactam derivative US Patent US10745401 Publication Date 8/18/2020 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2A |
|---|
| Name: | 5-hydroxytryptamine receptor 2A |
|---|
| Synonyms: | 5-HT-2 | 5-HT-2A | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT-2A) | 5-hydroxytryptamine receptor 2A (5HT-2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_HUMAN | HTR2 | HTR2A | Serotonin receptor 2A |
|---|
| Type: | undefined |
|---|
| Mol. Mass.: | 52607.65 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P28223 |
|---|
| Residue: | 471 |
|---|
| Sequence: | MDILCEENTSLSSTTNSLMQLNDDTRLYSNDFNSGEANTSDAFNWTVDSENRTNLSCEGC
LSPSCLSLLHLQEKNWSALLTAVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIAD
MLLGFLVMPVSMLTILYGYRWPLPSKLCAVWIYLDVLFSTASIMHLCAISLDRYVAIQNP
IHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSF
VSFFIPLTIMVITYFLTIKSLQKEATLCVSDLGTRAKLASFSFLPQSSLSSEKLFQRSIH
REPGSYTGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNEDVIGA
LLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENKKPLQLILVNTIPALAYK
SSQLQMGQKKNSKQDAKTTDNDCSMVALGKQHSEEASKDNSDGVNEKVSCV
|
|
|
|---|
| BDBM458718 |
|---|
| n/a |
|---|
| Name | BDBM458718 |
|---|
| Synonyms: | 5-{2-[4-(1,2-Benzoisoxazol-3-yl)piperidin-1-yl]ethyl}-6,7-dihydro[1,3]imidazolo[4,5-c]pyridin-4(5H)-one | US10745401, Example 191 | US11466007, Example 191 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H23N5O2 |
|---|
| Mol. Mass. | 365.4289 |
|---|
| SMILES | O=C1N(CCN2CCC(CC2)c2noc3ccccc23)CCc2[nH]cnc12 |
|---|
| Structure | 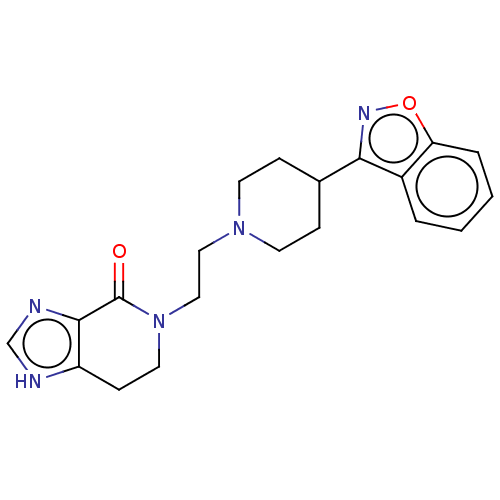
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yoshinaga, H; Ikuma, Y; Ikeda, J; Adachi, S; Mitsunuma, H; Aihara, Y; Besnard, J; Bell, AS Condensed lactam derivative US Patent US10745401 Publication Date 8/18/2020
Yoshinaga, H; Ikuma, Y; Ikeda, J; Adachi, S; Mitsunuma, H; Aihara, Y; Besnard, J; Bell, AS Condensed lactam derivative US Patent US10745401 Publication Date 8/18/2020