null
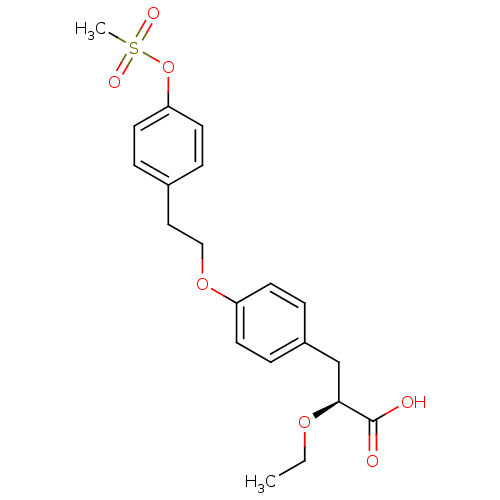
SMILES CCO[C@@H](Cc1ccc(OCCc2ccc(OS(C)(=O)=O)cc2)cc1)C(O)=O
InChI Key InChIKey=CXGTZJYQWSUFET-IBGZPJMESA-N
PDB links: 2 PDB IDs match this monomer.
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 28798
Found 4 hits for monomerid = 28798
TargetPeroxisome proliferator-activated receptor gamma(Homo sapiens (Human))
The University of Tokyo
Curated by ChEMBL
The University of Tokyo
Curated by ChEMBL
Affinity DataEC50: 360nMAssay Description:Ability to displace [3H]quipazine binding to 5-hydroxytryptamine 3 receptor sites in NG 108-15.More data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor alpha(Mus musculus)
The University of Tokyo
Curated by ChEMBL
The University of Tokyo
Curated by ChEMBL
Affinity DataEC50: 1.70E+3nMAssay Description:Compound was evaluated for its ability to displace [3H]quipazine binding to 5-hydroxytryptamine 3 receptor sites in NG 108-15 .More data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Mus musculus)
The University of Tokyo
Curated by ChEMBL
The University of Tokyo
Curated by ChEMBL
Affinity DataEC50: 250nMAssay Description:Ability to displace [3H]quipazine binding to 5-hydroxytryptamine 3 receptor sites in NG 108-15.More data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor alpha(Homo sapiens (Human))
The University of Tokyo
Curated by ChEMBL
The University of Tokyo
Curated by ChEMBL
Affinity DataEC50: 3.80E+3nMAssay Description:Ability to displace [3H]quipazine binding to 5-hydroxytryptamine 3 receptor sites in NG 108-15.More data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)