null
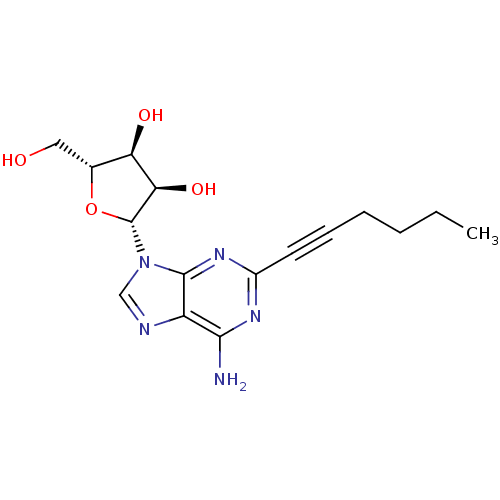
SMILES CCCCC#Cc1nc(N)c2ncn([C@@H]3O[C@H](CO)[C@@H](O)[C@H]3O)c2n1
InChI Key InChIKey=AHJRSKVOELIMOX-RVXWVPLUSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 7 hits for monomerid = 50119168
Found 7 hits for monomerid = 50119168
Affinity DataKi: 3.60nMAssay Description:Binding affinity against adenosine A2 receptor from rat striatal membranes with 50 nM CPA using [3H]-NECAMore data for this Ligand-Target Pair
Affinity DataKi: 8.10nMAssay Description:Binding affinity against adenosine A2 receptor from human platelet membranes using [3H]NECAMore data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:Inhibition of adenylate cyclase via adenosine A1 receptors in rat fat cell membranesMore data for this Ligand-Target Pair
Affinity DataKi: 16nMAssay Description:Binding affinity against low affinity component of adenosine A1 receptors from rat brain membranes using [3H]-DPCPXMore data for this Ligand-Target Pair
Affinity DataKi: 633nMAssay Description:Binding affinity against low affinity component of adenosine A1 receptors from rat brain membranes using [3H]-DPCPXMore data for this Ligand-Target Pair
Affinity DataEC50: 128nMAssay Description:Maximal NECA stimulation of adenylate cyclase via adenosine A2 receptor in human platelet membranesMore data for this Ligand-Target Pair
Affinity DataIC50: 2.30E+3nMAssay Description:Binding affinity against high affinity component of adenosine A1 receptors from rat brain membranes using [3H]DPCPXMore data for this Ligand-Target Pair