null
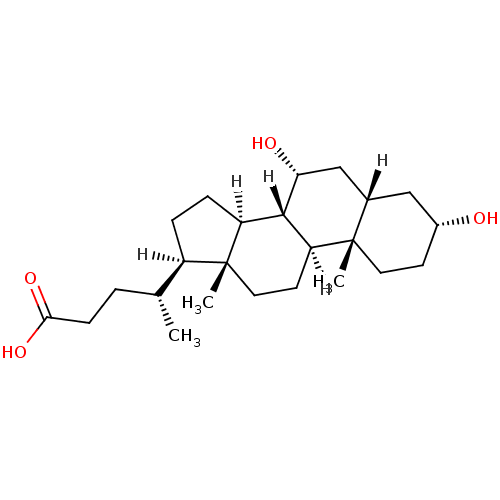
SMILES [H][C@@]1(CC[C@@]2([H])[C@]3([H])[C@H](O)C[C@]4([H])C[C@H](O)CC[C@]4(C)[C@@]3([H])CC[C@]12C)[C@H](C)CCC(O)=O
InChI Key InChIKey=RUDATBOHQWOJDD-BSWAIDMHSA-N
PDB links: 6 PDB IDs match this monomer. 34 PDB IDs contain this monomer as substructures. 34 PDB IDs contain inhibitors having a similarity of 90% to this monomer.
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 21674
Found 3 hits for monomerid = 21674
Affinity DataEC50: 8.66E+3nMAssay Description:Effective concentration against Farnesoid X receptor (FXR)More data for this Ligand-Target Pair
Affinity DataEC50: 1.30E+4nMAssay Description:Agonist activity at GST-tagged FXR-LBD using biotinylated-SRC-1 peptide as substrate preincubated with compound for 30 mins measured after 4 hrsMore data for this Ligand-Target Pair
Affinity DataEC50: 6.70E+3nMAssay Description:Agonist activity at TGR5 expressed in NCI-H716 cells assessed as cAMP level after 60 mins by FRET analysisMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)