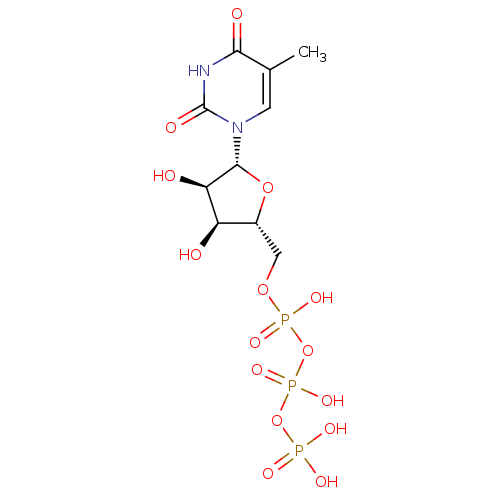
null
SMILES Cc1cn([C@@H]2O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]2O)c(=O)[nH]c1=O
InChI Key InChIKey=RZCIEJXAILMSQK-JXOAFFINSA-N
PDB links: 1 PDB ID matches this monomer.
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50205415
Found 3 hits for monomerid = 50205415
TargetP2Y purinoceptor 2(Homo sapiens (Human))
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
Affinity DataEC50: 480nMAssay Description:Agonist activity at human recombinant P2Y2 receptor expressed in 1321N1 cells assessed as stimulation of phospholipase CMore data for this Ligand-Target Pair
TargetP2Y purinoceptor 6(Homo sapiens (Human))
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
Affinity DataEC50: 140nMAssay Description:Agonist activity at human recombinant P2Y6 receptor expressed in 1321N1 cells assessed as stimulation of phospholipase CMore data for this Ligand-Target Pair
TargetP2Y purinoceptor 4(Homo sapiens (Human))
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
Affinity DataEC50: 3.90E+3nMAssay Description:Agonist activity at human recombinant P2Y4 receptor expressed in 1321N1 cells assessed as stimulation of phospholipase CMore data for this Ligand-Target Pair