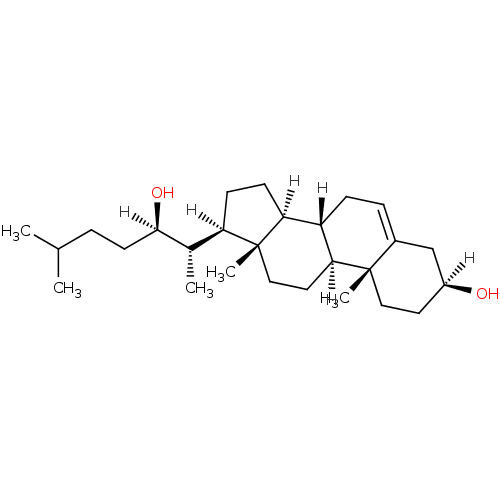
null
SMILES [H][C@@](O)(CCC(C)C)[C@@H](C)[C@@]1([H])CC[C@@]2([H])[C@]3([H])CC=C4C[C@@]([H])(O)CC[C@]4(C)[C@@]3([H])CC[C@]12C
InChI Key InChIKey=RZPAXNJLEKLXNO-GFKLAVDKSA-N
PDB links: 3 PDB IDs match this monomer.
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 20177
Found 5 hits for monomerid = 20177
Affinity DataEC50: 1.00E+3nMAssay Description:Agonist activity at human LXRalpha-LBD assessed as recruitment of co-activator peptide after 2 hrs by TR-FRET assayMore data for this Ligand-Target Pair
Affinity DataEC50: 325nMpH: 7.5 T: 2°CAssay Description:The LXR LiSA measures the ligand-dependent recruitment of a 25 amino acid fragment of the steroid receptor coactivator 1 (SRC1) to the ligand-binding...More data for this Ligand-Target Pair
Affinity DataEC50: >1.50E+4nMAssay Description:Agonist activity at human recombinant LXRalpha expressed in Escherichia coli BL21 cells assessed as association of recombinant SRC1 to LXRalpha ligan...More data for this Ligand-Target Pair
Affinity DataEC50: 1.50E+4nMAssay Description:Effective concentration for cofactor association with recombinant liver X receptor-alphaMore data for this Ligand-Target Pair
Affinity DataEC50: 6.71E+3nMAssay Description:Activation of GAL4 fused human LXR alpha expressed in HEK293 cells after 24 hrs by luciferase reporter gene assayMore data for this Ligand-Target Pair