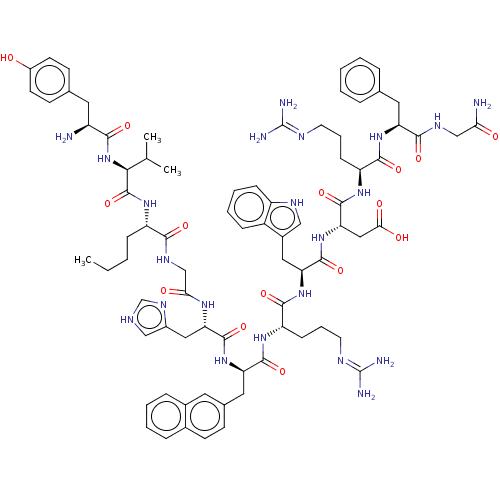
null
SMILES CCCC[C@H](NC(=O)[C@@H](NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(C)C)C(=O)NCC(=O)N[C@@H](Cc1c[nH]cn1)C(=O)N[C@H](Cc1ccc2ccccc2c1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccccc1)C(=O)NCC(N)=O
InChI Key InChIKey=WSDYWWDQJYPQDS-YREICWDXSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50134946
Found 3 hits for monomerid = 50134946
Affinity DataKi: 0.810nMAssay Description:Binding affinity against human Melanocortin 3 receptor by gamma-MCH displacement.More data for this Ligand-Target Pair
Affinity DataKi: 1nMAssay Description:Binding affinity against human Melanocortin 5 receptor by gamma-MCH displacement.More data for this Ligand-Target Pair
Affinity DataKi: 5.70nMAssay Description:Binding affinity against Melanocortin 4 receptor by gamma-MCH displacement.More data for this Ligand-Target Pair