null
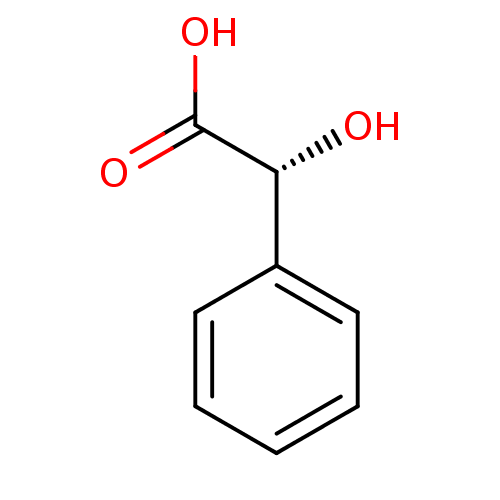
SMILES O[C@@H](C(O)=O)c1ccccc1
InChI Key InChIKey=IWYDHOAUDWTVEP-SSDOTTSWSA-N
PDB links: 5 PDB IDs match this monomer. 9 PDB IDs contain this monomer as substructures. 4 PDB IDs contain inhibitors having a similarity of 90% to this monomer.
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 16421
Found 5 hits for monomerid = 16421
Affinity DataKi: 3.61E+5nM ΔG°: -4.69kcal/molepH: 7.0 T: 2°CAssay Description:The production of NADPH from NADP+ and benzyl alcohol and xylitol was monitored by an increase in NADPH fluorescence (ex: 340 nm; em: 460 nm) using a...More data for this Ligand-Target Pair
TargetMandelate racemase(Pseudomonas putida (g-Proteobacteria))
Dalhousie University
Curated by ChEMBL
Dalhousie University
Curated by ChEMBL
Affinity DataKi: 5.90E+5nMpH: 7.5Assay Description:Inhibition constant against Mandelate racemase from Pseudomonas putida at pH 7.5More data for this Ligand-Target Pair
TargetMandelate racemase(Pseudomonas putida (g-Proteobacteria))
Dalhousie University
Curated by ChEMBL
Dalhousie University
Curated by ChEMBL
Affinity DataKi: 8.10E+5nMAssay Description:Inhibition of mandelate racemase in Pseudomonas putidaMore data for this Ligand-Target Pair
TargetTyrosine-protein phosphatase non-receptor type 7(Homo sapiens (Human))
Burnham Center for Chemical Genomics
Curated by PubChem BioAssay
Burnham Center for Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 1.00E+5nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetTyrosine-protein phosphatase non-receptor type 1(Homo sapiens (Human))
Brown University
Curated by ChEMBL
Brown University
Curated by ChEMBL
Affinity DataIC50: 5.50E+6nMAssay Description:Inhibitory activity against Yersinia Protein-tyrosine phosphatase 1BMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)