null
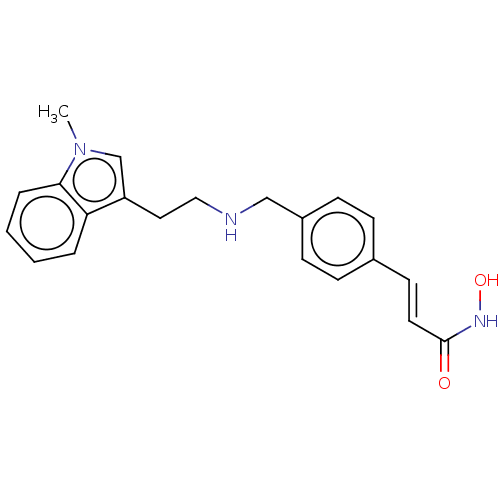
SMILES Cn1cc(CCNCc2ccc(\C=C\C(=O)NO)cc2)c2ccccc12
InChI Key InChIKey=AEDUBEGTPWKDCX-ZHACJKMWSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 19 hits for monomerid = 323704
Found 19 hits for monomerid = 323704
Affinity DataIC50: 0.200nMAssay Description:All histone deacetylases were purchased from BPS Bioscience. The substrates, Broad Substrate A, and Broad Substrate B, were synthesized and are now a...More data for this Ligand-Target Pair
In DepthDetails
Affinity DataIC50: 0.400nMAssay Description:All histone deacetylases were purchased from BPS Bioscience. The substrates, Broad Substrate A, and Broad Substrate B, were synthesized and are now a...More data for this Ligand-Target Pair
Affinity DataIC50: 655nMAssay Description:All histone deacetylases were purchased from BPS Bioscience. The substrates, Broad Substrate A, and Broad Substrate B, were synthesized and are now a...More data for this Ligand-Target Pair
Affinity DataIC50: 99nMAssay Description:All histone deacetylases were purchased from BPS Bioscience. The substrates, Broad Substrate A, and Broad Substrate B, were synthesized and are now a...More data for this Ligand-Target Pair
Affinity DataIC50: 1.60nMAssay Description:All histone deacetylases were purchased from BPS Bioscience. The substrates, Broad Substrate A, and Broad Substrate B, were synthesized and are now a...More data for this Ligand-Target Pair
Affinity DataIC50: 2.04E+3nMAssay Description:All histone deacetylases were purchased from BPS Bioscience. The substrates, Broad Substrate A, and Broad Substrate B, were synthesized and are now a...More data for this Ligand-Target Pair
Affinity DataIC50: 1.04E+3nMAssay Description:All histone deacetylases were purchased from BPS Bioscience. The substrates, Broad Substrate A, and Broad Substrate B, were synthesized and are now a...More data for this Ligand-Target Pair
Affinity DataIC50: 4.85E+3nMAssay Description:All histone deacetylases were purchased from BPS Bioscience. The substrates, Broad Substrate A, and Broad Substrate B, were synthesized and are now a...More data for this Ligand-Target Pair
TargetHistone deacetylase(Homo sapiens (Human))
Novartis Institute for Biomedical Research
Curated by ChEMBL
Novartis Institute for Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 23nMAssay Description:Inhibitory activity against histone deacetylase (HDAC) enzyme obtained from H1299 human lung carcinoma cell lysatesMore data for this Ligand-Target Pair
Target InfoPDBMMDBNCI pathwayReactome pathwayKEGG
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
In DepthDetails
Target InfoPDBMMDBNCI pathwayReactome pathwayKEGG
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
In DepthDetails
Target InfoPDBNCI pathwayReactome pathwayKEGG
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
In DepthDetails
In DepthDetails
In DepthDetails
Target InfoPDBMMDBNCI pathwayReactome pathwayKEGG
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
In DepthDetails
Target InfoPDBMMDBNCI pathwayReactome pathwayKEGG
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
In DepthDetails
Target InfoPDBMMDBNCI pathwayReactome pathwayKEGG
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
In DepthDetails
Affinity DataIC50: 1.20nMAssay Description:All histone deacetylases were purchased from BPS Bioscience. The substrates, Broad Substrate A, and Broad Substrate B, were synthesized and are now a...More data for this Ligand-Target Pair