null
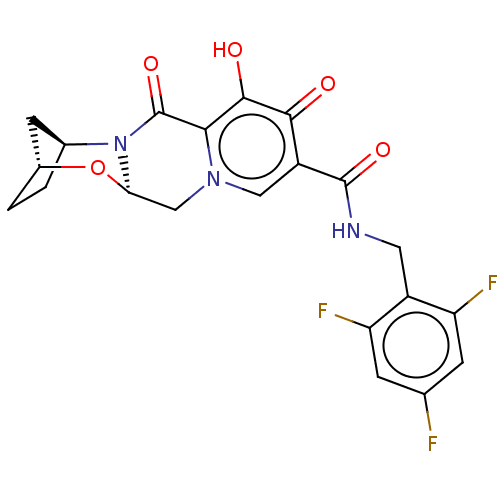
SMILES Oc1c2C(=O)N3[C@H]4CC[C@H](C4)O[C@@H]3Cn2cc(C(=O)NCc2c(F)cc(F)cc2F)c1=O
InChI Key InChIKey=SOLUWJRYJLAZCX-LYOVBCGYSA-N
PDB links: 4 PDB IDs match this monomer.
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 330048
Found 6 hits for monomerid = 330048
Affinity DataKi: 3.02E+5nMAssay Description:This is a review article. Please point to the original journal.More data for this Ligand-Target Pair
TargetPOU domain, class 2, transcription factor 2(Homo sapiens (Human))
Gilead Sciences, Inc.
US Patent
Gilead Sciences, Inc.
US Patent
Affinity DataIC50: 487nMAssay Description:The dose dependent inhibition of OCT2 mediated uptake of a model substrate 14C-Tetraethylammonium (TEA) by test compounds was studied in wild-type an...More data for this Ligand-Target Pair
Affinity DataIC50: 487nMAssay Description:The dose dependent inhibition of OCT2 mediated uptake of a model substrate 14C-Tetraethylammonium (TEA) by test compounds was studied in wild-type an...More data for this Ligand-Target Pair
Affinity DataIC50: 487nMAssay Description:The dose dependent inhibition of OCT2 mediated uptake of a model substrate 14C-Tetraethylammonium (TEA) by test compounds was studied in wild-type an...More data for this Ligand-Target Pair
Affinity DataIC50: 7.5nMAssay Description:Inhibition of HIV integrase strand transfer activityMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)