null
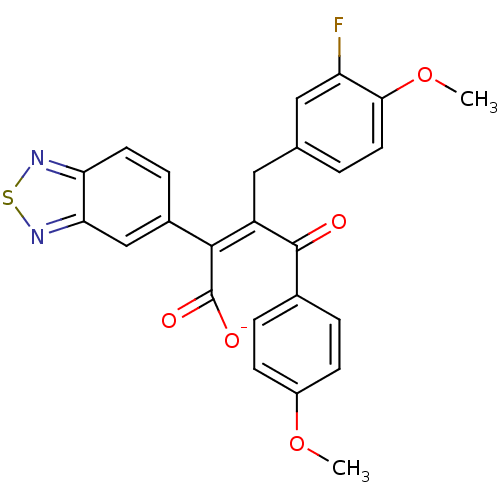
SMILES COc1ccc(cc1)C(=O)C(\Cc1ccc(OC)c(F)c1)=C(/C([O-])=O)c1ccc2nsnc2c1
InChI Key InChIKey=WPTYOJVFRZMDBQ-NKFKGCMQSA-M
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 50070879
Found 2 hits for monomerid = 50070879
Affinity DataIC50: 14nMAssay Description:In vitro ability to inhibit specific [125I]ET1 binding to rat aorta membranes Endothelin A receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 2.90E+3nMAssay Description:In vitro ability to inhibit specific [125I]ET1 binding to porcine kidney (inner medulla) membranes Endothelin B receptorMore data for this Ligand-Target Pair