null
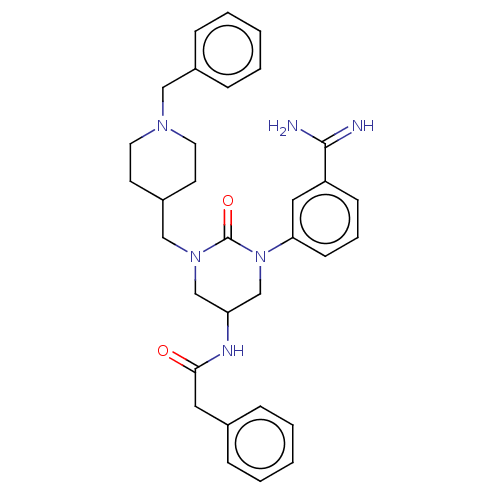
SMILES NC(=N)c1cccc(c1)N1CC(CN(CC2CCN(Cc3ccccc3)CC2)C1=O)NC(=O)Cc1ccccc1
InChI Key InChIKey=BFOHAEDJCBXVHR-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 50158153
Found 6 hits for monomerid = 50158153
Affinity DataKi: 380nMAssay Description:Inhibition of human recombinant hepatocyte growth factor activator using H2N(EEdansyl)GKQLRVVNGG (KDabcyl)-NH2 as substrate by fluorometric analysisMore data for this Ligand-Target Pair
Affinity DataKi: 500nMAssay Description:Compound was evaluated for potency towards nicotinic acetylcholine receptor in rat P2 brain membranes using [3H]-nicotine as a radioligandMore data for this Ligand-Target Pair
Affinity DataKi: 740nMAssay Description:Inhibition of human recombinant factor 10a using H2N(EEdansyl)GKQLRVVNGG (KDabcyl)-NH2 as substrate by fluorometric analysisMore data for this Ligand-Target Pair
Affinity DataKi: 1.12E+3nMAssay Description:Inhibition of human recombinant hepsin using H2N(EEdansyl)GKQLRVVNGG (KDabcyl)-NH2 as substrate by fluorometric analysisMore data for this Ligand-Target Pair
TargetSuppressor of tumorigenicity 14 protein(Homo sapiens (Human))
Southern Research
Curated by ChEMBL
Southern Research
Curated by ChEMBL
Affinity DataKi: 1.28E+3nMAssay Description:Inhibition of human recombinant matripase catalytic domain using H2N(EEdansyl)GKQLRVVNGG (KDabcyl)-NH2 as substrate by fluorometric analysisMore data for this Ligand-Target Pair
Affinity DataKi: 4.23E+4nMAssay Description:Inhibition of human recombinant thrombin using H2N(EEdansyl)GKQLRVVNGG (KDabcyl)-NH2 as substrate by fluorometric analysisMore data for this Ligand-Target Pair