null
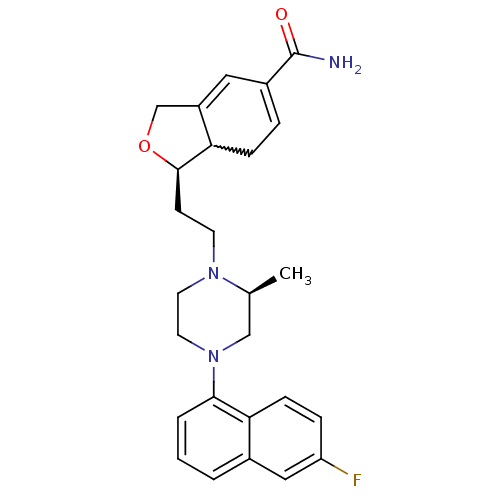
SMILES C[C@H]1CN(CCN1CC[C@H]1OCC2=CC(=CCC12)C(N)=O)c1cccc2cc(F)ccc12
InChI Key InChIKey=TZGHJRPUBWXMCC-PBGLVDLSSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50210634
Found 5 hits for monomerid = 50210634
Affinity DataKi: 8.80nMAssay Description:Displacement of [3H]citalopram from human 5HT transpoter expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 13nMAssay Description:Displacement of [3H]8-OH-DPAT from human 5HT1A expressed in LM(tk-) cellsMore data for this Ligand-Target Pair
Affinity DataKi: 67nMAssay Description:Displacement of [3H]GR125743 from human 5HT1D expressed in LM(tk-) cellsMore data for this Ligand-Target Pair
Affinity DataKi: 107nMAssay Description:Displacement of [3H]raclopride from dopamine D2 receptor in rat caudate membranesMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Displacement of [3H]GR125743 from human 5HT1B expressed in LM(tk-) cellsMore data for this Ligand-Target Pair