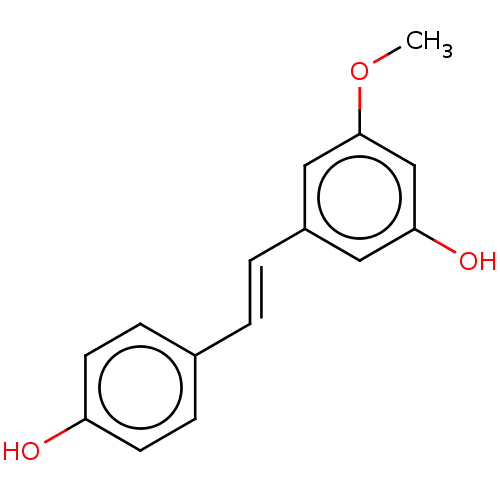
null
SMILES COc1cc(O)cc(\C=C\c2ccc(O)cc2)c1
InChI Key InChIKey=KUWZXOMQXYWKBS-NSCUHMNNSA-N
PDB links: 3 PDB IDs match this monomer.
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50252422
Found 4 hits for monomerid = 50252422
TargetCytochrome P450 1A1(Homo sapiens (Human))
Laboratory of Medicinal Chemistry, Endocrinology and Nephrology Unit, CHU de Québec - Research Center, Québec, Québec, Canada; Department of Molecular Medicine, Faculty of Medicine, Université Laval,
Curated by ChEMBL
Laboratory of Medicinal Chemistry, Endocrinology and Nephrology Unit, CHU de Québec - Research Center, Québec, Québec, Canada; Department of Molecular Medicine, Faculty of Medicine, Université Laval,
Curated by ChEMBL
Affinity DataKi: 130nMAssay Description:Inhibition of recombinant human CYP1A1 expressed in supersomes coexpressing NADPH-CYP reductase using 7-ethoxyresorufin as substrate after 15 mins in...More data for this Ligand-Target Pair
TargetCytochrome P450 1B1(Homo sapiens (Human))
Laboratory of Medicinal Chemistry, Endocrinology and Nephrology Unit, CHU de Québec - Research Center, Québec, Québec, Canada; Department of Molecular Medicine, Faculty of Medicine, Université Laval,
Curated by ChEMBL
Laboratory of Medicinal Chemistry, Endocrinology and Nephrology Unit, CHU de Québec - Research Center, Québec, Québec, Canada; Department of Molecular Medicine, Faculty of Medicine, Université Laval,
Curated by ChEMBL
Affinity DataKi: 900nMAssay Description:Inhibition of recombinant human CYP1B1 expressed in supersomes coexpressing NADPH-CYP reductase using 7-ethoxyresorufin as substrate after 15 mins in...More data for this Ligand-Target Pair
Target InfoPDBMMDBNCI pathwayReactome pathwayKEGG
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
UniProtKB/SwissProt
B.MOADDrugBankantibodypediaGoogleScholar
In DepthDetails
In DepthDetails