null
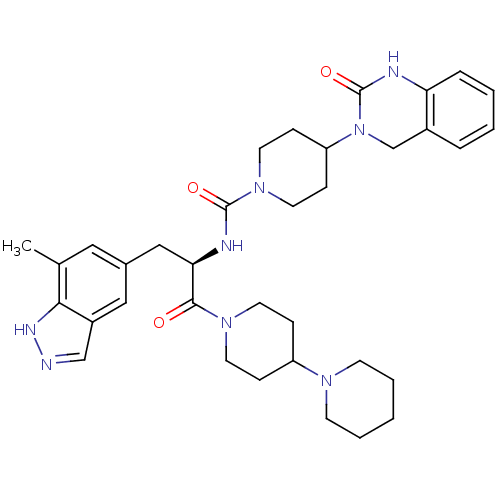
SMILES Cc1cc(C[C@@H](NC(=O)N2CCC(CC2)N2Cc3ccccc3NC2=O)C(=O)N2CCC(CC2)N2CCCCC2)cc2cn[nH]c12
InChI Key InChIKey=MMNFZCWXLBJTKN-WJOKGBTCSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 14 hits for monomerid = 50273292
Found 14 hits for monomerid = 50273292
TargetCalcitonin gene-related peptide type 1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Affinity DataKi: 0.00730nMAssay Description:Binding affinity to CGRP receptor (unknown origin)More data for this Ligand-Target Pair
TargetCalcitonin gene-related peptide type 1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Affinity DataKi: 0.0100nMAssay Description:Displacement of [I125]CGRP from human CGRP receptor in SK-N-MC cellsMore data for this Ligand-Target Pair
TargetCalcitonin gene-related peptide type 1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Affinity DataKi: 0.0120nMAssay Description:Binding affinity to CGRP receptor (unknown origin)More data for this Ligand-Target Pair
TargetCalcitonin gene-related peptide type 1 receptor(Homo sapiens (Human))
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Affinity DataEC50: 0.0170nMAssay Description:Antagonist activity at CGRP receptor in human SK-N-MC cells assessed as inhibition of CCRP-induced cAMP productionMore data for this Ligand-Target Pair
TargetCytochrome P450 3A4(Homo sapiens (Human))
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Affinity DataIC50: 6.00E+3nMAssay Description:Inhibition of CYP3A4 (unknown origin) using 7-benzyloxy-resorufin as substrateMore data for this Ligand-Target Pair
TargetCytochrome P450 3A4(Homo sapiens (Human))
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Affinity DataIC50: 1.10E+3nMAssay Description:Inhibition of CYP3A4 (unknown origin) using 7-benzyloxy-resorufin as substrateMore data for this Ligand-Target Pair
TargetCytochrome P450 2D6(Homo sapiens (Human))
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Affinity DataIC50: 4.00E+4nMAssay Description:Inhibition of CYP2D6 (unknown origin)More data for this Ligand-Target Pair
TargetCytochrome P450 2C9(Homo sapiens (Human))
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Affinity DataIC50: 4.00E+4nMAssay Description:Inhibition of CYP2C9 (unknown origin)More data for this Ligand-Target Pair
TargetCytochrome P450 2C19(Homo sapiens (Human))
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Affinity DataIC50: 4.00E+4nMAssay Description:Inhibition of CYP2C19 (unknown origin)More data for this Ligand-Target Pair
TargetCytochrome P450 1A2(Homo sapiens (Human))
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Affinity DataIC50: 4.00E+4nMAssay Description:Inhibition of CYP1A2 (unknown origin)More data for this Ligand-Target Pair
TargetCytochrome P450 3A4(Homo sapiens (Human))
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Affinity DataIC50: 3.60E+4nMAssay Description:Inhibition of CYP3A4 (unknown origin) using 7-benzyloxy-4-trifluoromethylcoumarin as substrateMore data for this Ligand-Target Pair
TargetCytochrome P450 3A4(Homo sapiens (Human))
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Affinity DataIC50: 6.00E+3nMAssay Description:Inhibition of recombinant CYP3A4 (unknown origin) expressed in insect microsomes after 20 mins in presence of BFC substrateMore data for this Ligand-Target Pair
TargetCytochrome P450 3A4(Homo sapiens (Human))
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Affinity DataIC50: 3.60E+4nMAssay Description:Inhibition of recombinant CYP3A4 (unknown origin) expressed in insect microsomes after 45 mins in presence of BZR substrateMore data for this Ligand-Target Pair
TargetCytochrome P450 3A4(Homo sapiens (Human))
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
Affinity DataIC50: 2.40E+4nMAssay Description:Inhibition of CYP3A4 (unknown origin) using 7-benzyloxy-4-trifluoromethylcoumarin as substrateMore data for this Ligand-Target Pair