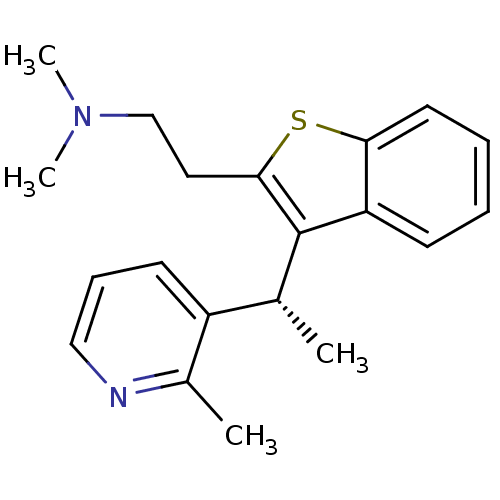
null
SMILES C[C@@H](c1c(CCN(C)C)sc2ccccc12)c1cccnc1C
InChI Key InChIKey=HOUUGSUKNBXWNT-CQSZACIVSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50314273
Found 5 hits for monomerid = 50314273
Affinity DataKi: 17nMAssay Description:Displacement of [3H]pyrilamine from human histamine H1 receptor expressed in CHO Flp-In cells after 90 mins by scintillation countingMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Neurocrine Biosciences
Curated by ChEMBL
Neurocrine Biosciences
Curated by ChEMBL
Affinity DataKi: >5.00E+3nMAssay Description:Displacement of [3H]Dofetilide from human ERGMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M1(Homo sapiens (Human))
Neurocrine Biosciences
Curated by ChEMBL
Neurocrine Biosciences
Curated by ChEMBL
Affinity DataKi: >1.00E+4nMAssay Description:Displacement of [3H]N-methylscopolamine from human muscarinic M1 receptor expressed in CHO Flp-In cells after 90 mins by scintillation countingMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of recombinant CYP3A4 after 30 mins by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of recombinant CYP2D6 after 30 mins by fluorescence assayMore data for this Ligand-Target Pair