null
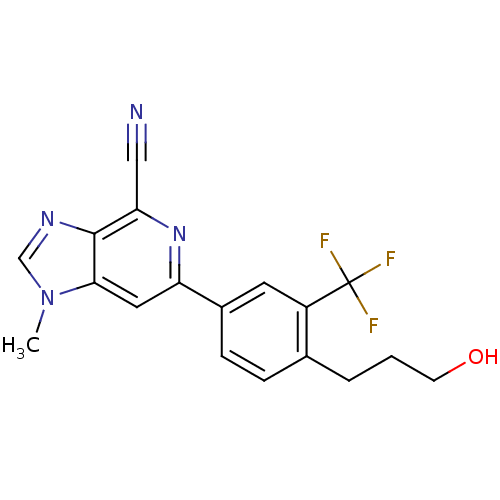
SMILES Cn1cnc2c(nc(cc12)-c1ccc(CCCO)c(c1)C(F)(F)F)C#N
InChI Key InChIKey=JDJQLDQXYPUJRN-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50323258
Found 3 hits for monomerid = 50323258
Affinity DataIC50: 234nMAssay Description:The inhibitory activity of the compounds of the invention was demonstrated in vitro by measuring the inhibition of recombinant human Cathepsin S as f...More data for this Ligand-Target Pair
Affinity DataIC50: 24nMAssay Description:Inhibition of human cathepsin S by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.07E+3nMAssay Description:Inhibition of human cathepsin K by fluorescence assayMore data for this Ligand-Target Pair