null
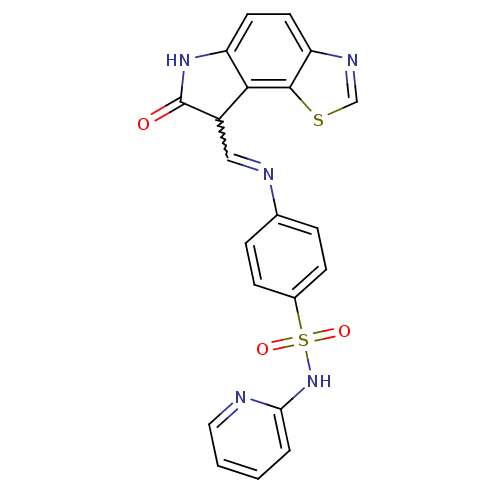
SMILES O=C1Nc2ccc3ncsc3c2C1C=Nc1ccc(cc1)S(=O)(=O)Nc1ccccn1
InChI Key InChIKey=SIPNIVSCAYGONU-UHFFFAOYSA-N
PDB links: 1 PDB ID matches this monomer.
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 7 hits for monomerid = 7755
Found 7 hits for monomerid = 7755
Affinity DataIC50: 100nMAssay Description:The biochemical activity of compounds was determined by incubation with specific enzymes and substrates in the presence 1.4 uM ATP/[gamma-32P] ATP. A...More data for this Ligand-Target Pair
Affinity DataIC50: 9.70nMAssay Description:The biochemical activity of compounds was determined by incubation with specific enzymes and substrates in the presence 1.4 uM ATP/[gamma-32P] ATP. A...More data for this Ligand-Target Pair
TargetPutative glycogen synthase kinase 3 alpha(Ustilago maydis (Smut fungus))
Technische Universität Dortmund
Technische Universität Dortmund
Affinity DataIC50: 2.20E+3nMAssay Description:Inhibition of UmGSK3 by kinase inhibitors.More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:Inhibition of Cyclin-dependent kinase 2More data for this Ligand-Target Pair
TargetHigh affinity nerve growth factor receptor(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataIC50: 9nMAssay Description:Inhibition of receptor Tyrosine kinase A, TrkA (nerve growth factor receptor)More data for this Ligand-Target Pair
Affinity DataIC50: 9.70nMAssay Description:Evaluated for inhibition of human cyclin dependent kinase 2 (CDK2)More data for this Ligand-Target Pair
TargetCyclin-A1/Cyclin-A2/Cyclin-dependent kinase 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of Cyclin-dependent kinase 2 cyclin AMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)