null
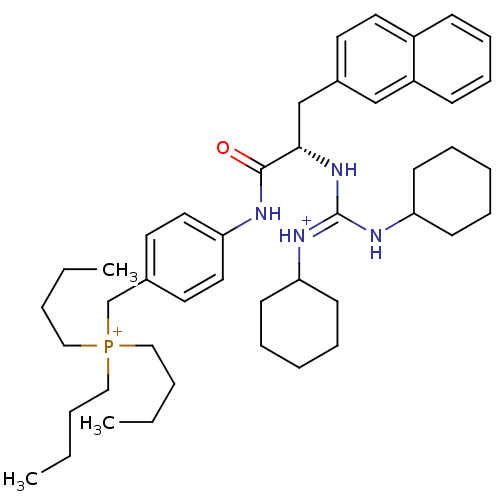
SMILES CCCC[P+](CCCC)(CCCC)Cc1ccc(NC(=O)[C@H](Cc2ccc3ccccc3c2)N\C(NC2CCCCC2)=[NH+]\C2CCCCC2)cc1
InChI Key InChIKey=UKMJWGFHXMGRNG-QLKFWGTOSA-P
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50045219
Found 4 hits for monomerid = 50045219
TargetB2 bradykinin receptor(Homo sapiens (Human))
Sterling Winthrop Pharmaceutical Research Division
Curated by ChEMBL
Sterling Winthrop Pharmaceutical Research Division
Curated by ChEMBL
Affinity DataKi: 60nMAssay Description:Binding affinity against human bradykinin receptor B2 using [3H]bradykinin as radioligandMore data for this Ligand-Target Pair
TargetB2 bradykinin receptor(Homo sapiens (Human))
Sterling Winthrop Pharmaceutical Research Division
Curated by ChEMBL
Sterling Winthrop Pharmaceutical Research Division
Curated by ChEMBL
Affinity DataKi: 60nMAssay Description:Binding affinity against human bradykinin receptor B2 using [3H]bradykinin as radioligandMore data for this Ligand-Target Pair
TargetB2 bradykinin receptor(Homo sapiens (Human))
Sterling Winthrop Pharmaceutical Research Division
Curated by ChEMBL
Sterling Winthrop Pharmaceutical Research Division
Curated by ChEMBL
Affinity DataKi: 60nMAssay Description:Binding affinity against human IMR90 fetal lung fibroblast bradykinin B2 receptor was evaluatedMore data for this Ligand-Target Pair
TargetMuscarinic acetylcholine receptor M1/M2/M3/M4/M5(RAT)
Sterling Winthrop Pharmaceutical Research Division
Curated by ChEMBL
Sterling Winthrop Pharmaceutical Research Division
Curated by ChEMBL
Affinity DataKi: 350nMAssay Description:Binding affinity against rat muscarinic receptorMore data for this Ligand-Target Pair