null
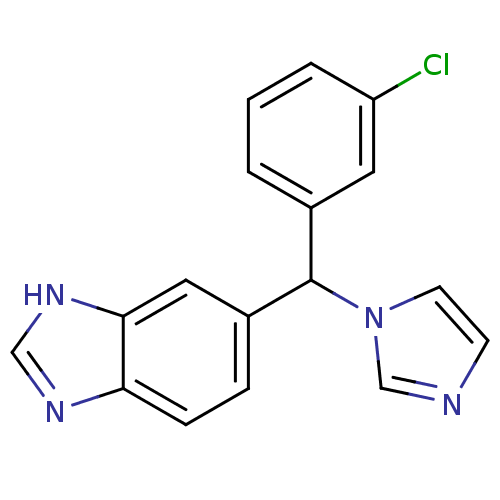
SMILES Clc1cccc(c1)C(c1ccc2nc[nH]c2c1)n1ccnc1
InChI Key InChIKey=UGFHIPBXIWJXNA-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 13 hits for monomerid = 50176808
Found 13 hits for monomerid = 50176808
Affinity DataEC50: 7.00E+3nMAssay Description:The imidazole derivatives were evaluated for their retinoic acid metabolism inhibitory activity using a MCF-7 cell assay, using radiolabelled all-tra...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:A commercially available P450-GLO Assay kit (Promega Corporation, Madison Wis.) is used to screen various compounds for CYP3A4A inhibition activity. ...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:Cytochrome P450 is a large and diverse group of enzymes that catalyze the oxidation of organic substances. Some members of the CYP family contribute ...More data for this Ligand-Target Pair
Target1,25-dihydroxyvitamin D(3) 24-hydroxylase, mitochondrial(Homo sapiens (Human))
Cardiff University
Curated by ChEMBL
Cardiff University
Curated by ChEMBL
Affinity DataIC50: 2.30nMAssay Description:Inhibition of human MBP-tagged CYP24A1 expressed in Escherichia coli using 1,25(OH)2D3 substrate in presence of bovine adrenodoxin, adrenodoxin reduc...More data for this Ligand-Target Pair
Affinity DataIC50: 6.00E+3nMAssay Description:Potency towards cytochrome P 450 26 enzyme activityMore data for this Ligand-Target Pair
Affinity DataIC50: 2.45E+3nMAssay Description:Inhibition of CYP26A1 in ATRA-induced human HL60 cell microsomes incubated for 30 mins using ATRA and NADPH by HPLC methodMore data for this Ligand-Target Pair
Affinity DataIC50: 7.00E+3nMAssay Description:Inhibition of CYP26A1 in human MCF7 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 690nMAssay Description:Inhibition of CYP3A4 (unknown origin) incubated for 45 mins using NADPH by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 4.76E+3nMAssay Description:Inhibition of CYP2D6 (unknown origin) incubated for 45 mins using NADPH and ATRA by HPLC assayMore data for this Ligand-Target Pair
Affinity DataIC50: 540nMAssay Description:Inhibition of CYP26A1 in human MCF7 cell microsomes using [3H]ATRA after 1 hr by scintillation countingMore data for this Ligand-Target Pair
Affinity DataIC50: 540nMAssay Description:Inhibition of human CYP26A1 assessed using [11,12-3H]ATRA as substrate by scintillation countingMore data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Inhibition of human placental microsome CYP19More data for this Ligand-Target Pair
Affinity DataIC50: 890nMAssay Description:Inhibition of CYP26A1 in ATRA-induced human HL60 cell microsomes incubated for 30 mins in dark condition with NADPH and ATRA by HPLC methodMore data for this Ligand-Target Pair