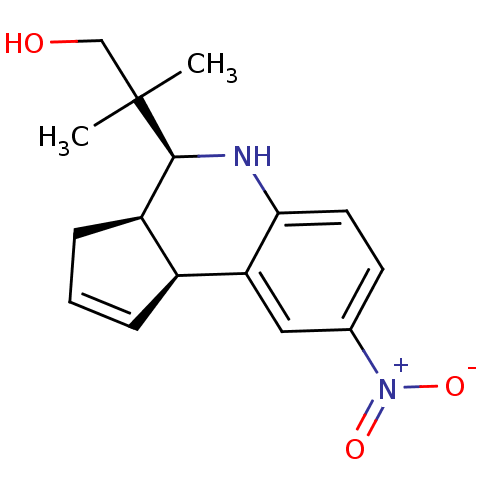
null
SMILES CC(C)(CO)[C@H]1Nc2ccc(cc2[C@H]2C=CC[C@@H]12)[N+]([O-])=O
InChI Key InChIKey=CJRZFRLQNHKDCY-ZOWXZIJZSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 8 hits for monomerid = 50338693
Found 8 hits for monomerid = 50338693
Affinity DataIC50: 1.80E+4nMAssay Description:Displacement of [3H]dexamethasone from Sprague-Dawley rat GR by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataIC50: 6.30E+3nMAssay Description:Displacement of [3H]aldosterone from Sprague-Dawley rat MR by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataIC50: 13nMAssay Description:Displacement of [3H]testosterone from Sprague-Dawley rat AR by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataIC50: 5.10nMAssay Description:Competitive binding affinity to rat androgen receptorChecked by AuthorMore data for this Ligand-Target Pair
Affinity DataEC50: 2.30nMAssay Description:Agonist activity at rat androgen receptorChecked by AuthorMore data for this Ligand-Target Pair
Affinity DataIC50: 130nMAssay Description:Displacement of [3H]progesterone from rabbit PR by liquid scintillation countingMore data for this Ligand-Target Pair