null
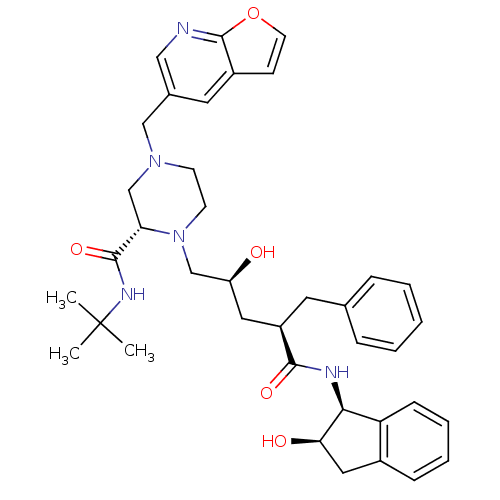
SMILES CC(C)(C)NC(=O)[C@@H]1CN(Cc2cnc3occc3c2)CCN1C[C@@H](O)C[C@@H](Cc1ccccc1)C(=O)N[C@@H]1[C@H](O)Cc2ccccc12
InChI Key InChIKey=UBUFVRJUGFJQSI-PPJSLLJVSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50408997
Found 3 hits for monomerid = 50408997
Affinity DataKi: 7.50E+3nMAssay Description:Mechanism based inhibition of human cytochrome P450 3A4 measured by testosterone 6-beta hydroxylationMore data for this Ligand-Target Pair
Affinity DataKi: 3.20E+4nMAssay Description:Mechanism based inhibition of human cytochrome P450 2D6 measured by bufurarol 1'-hydroxylationMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 0.350nMAssay Description:Inhibition of HIV-1 protease in vitro.More data for this Ligand-Target Pair