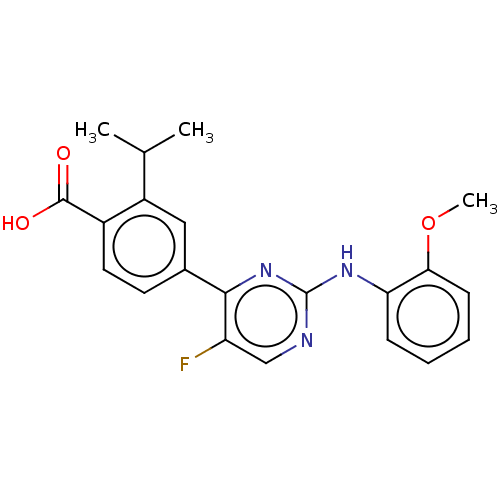
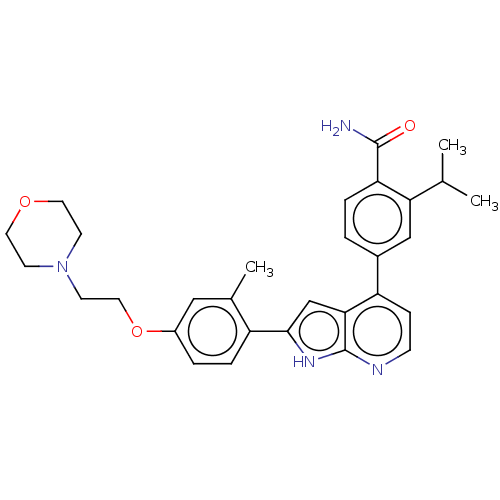
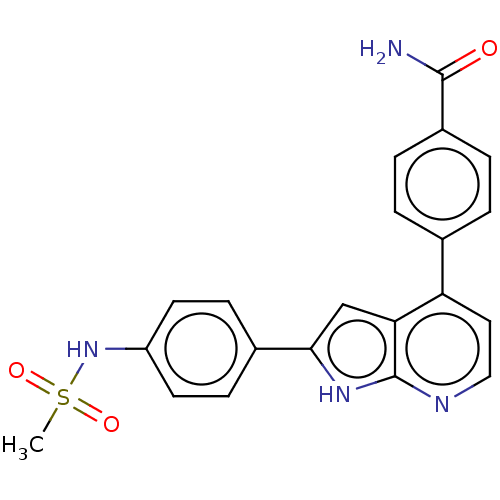
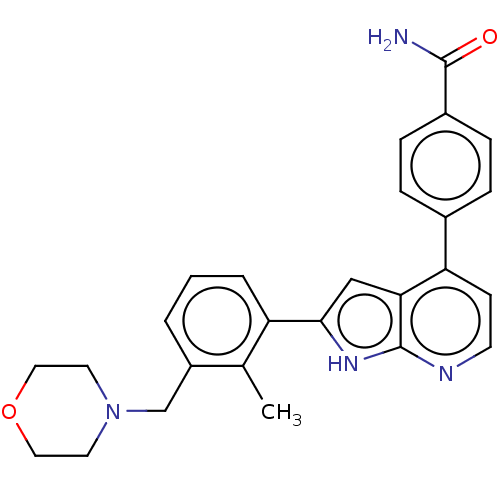
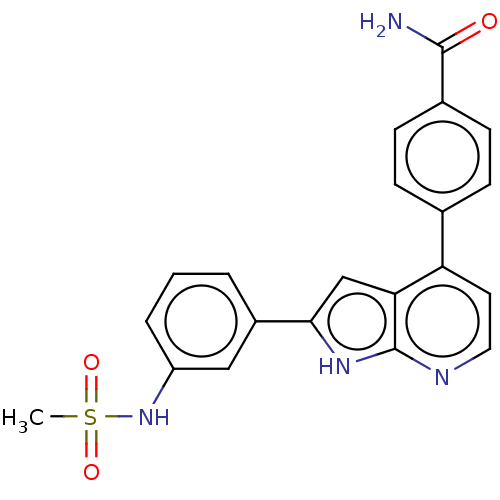
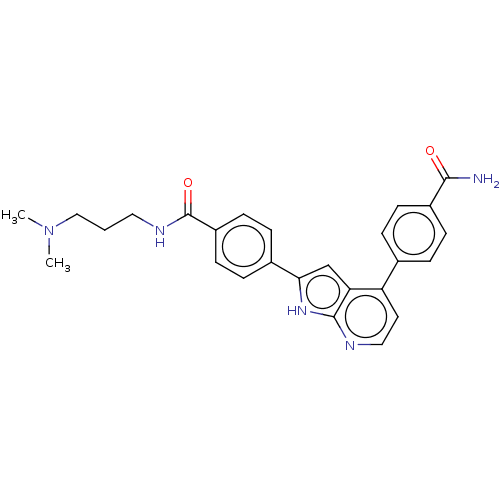
Affinity DataKi: 0.135nMAssay Description:Displacement of [N-methyl-3H]nisoxetine from rat hippocampus NET by filtration binding assayMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(MOUSE)
GlaxoSmithKline Medicine Research Centre
Curated by ChEMBL
GlaxoSmithKline Medicine Research Centre
Curated by ChEMBL
Affinity DataKi: 0.851nMAssay Description:Displacement of [3H]citalopram from mouse cortex SERT by filtration binding assayMore data for this Ligand-Target Pair
TargetSodium-dependent noradrenaline transporter(MOUSE)
GlaxoSmithKline Medicines Research Centre
Curated by ChEMBL
GlaxoSmithKline Medicines Research Centre
Curated by ChEMBL
Affinity DataKi: 1.58nMAssay Description:Displacement of [3H]nisoxetine from NET in mouse brainMore data for this Ligand-Target Pair
TargetSodium-dependent serotonin transporter(MOUSE)
GlaxoSmithKline Medicine Research Centre
Curated by ChEMBL
GlaxoSmithKline Medicine Research Centre
Curated by ChEMBL
Affinity DataKi: 1.58nMAssay Description:Displacement of [3H]-citalopram from SERT in mouse cortex after 2 hrs by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataKi: 2.51nMAssay Description:Displacement of [3H]nisoxetine from NET in rat hippocampus after 2 hrs by liquid scintillation countingMore data for this Ligand-Target Pair
TargetSodium-dependent dopamine transporter(MOUSE)
GlaxoSmithKline Medicines Research Centre
Curated by ChEMBL
GlaxoSmithKline Medicines Research Centre
Curated by ChEMBL
Affinity DataKi: 10nMAssay Description:Displacement of [3H]WIN-35428 from DAT in mouse brainMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 25.1nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 63.1nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 200nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 200nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 398nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 398nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 501nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 501nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 501nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 631nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 794nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetHistamine H1 receptor(Homo sapiens (Human))
GlaxoSmithKline Medicine Research Centre
Curated by ChEMBL
GlaxoSmithKline Medicine Research Centre
Curated by ChEMBL
Affinity DataKi: 1.00E+3nMAssay Description:Inhibition of histamine H1 receptorMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 1.00E+3nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 1.00E+3nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 1.00E+3nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 1.26E+3nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 1.58E+3nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 2.51E+3nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 2.51E+3nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 2.51E+3nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 2.51E+3nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 3.98E+3nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 3.98E+3nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 6.31E+3nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: >7.94E+3nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 7.94E+3nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 1.26E+4nMAssay Description:from Dfrom ifrom sfrom pfrom lfrom afrom cfrom efrom mfrom efrom nfrom tfrom from ofrom ffrom from [from 3from Hfrom ]from dfrom ofrom ffrom efrom tf...More data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 1.26E+4nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 3.16E+4nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataKi: 3.16E+4nMAssay Description:Displacement of [3H]-dofetilide from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
TargetCalcium/calmodulin-dependent protein kinase kinase 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataIC50: 1.60nMAssay Description:Inhibition of full-length human GST-tagged CAMKK2 using 5FAM-AKPKGNKDYHLQTCCGSLAYRRR-amide as substrate preincubated for 30 mins followed by substrat...More data for this Ligand-Target Pair
TargetCalcium/calmodulin-dependent protein kinase kinase 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataIC50: 2nMAssay Description:Inhibition of full-length human GST-tagged CAMKK2 using 5FAM-AKPKGNKDYHLQTCCGSLAYRRR-amide as substrate preincubated for 30 mins followed by substrat...More data for this Ligand-Target Pair
TargetCalcium/calmodulin-dependent protein kinase kinase 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataIC50: 2nMAssay Description:Inhibition of full-length human GST-tagged CAMKK2 using 5FAM-AKPKGNKDYHLQTCCGSLAYRRR-amide as substrate preincubated for 30 mins followed by substrat...More data for this Ligand-Target Pair
TargetCalcium/calmodulin-dependent protein kinase kinase 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataIC50: 4nMAssay Description:Inhibition of full-length human GST-tagged CAMKK2 using 5FAM-AKPKGNKDYHLQTCCGSLAYRRR-amide as substrate preincubated for 30 mins followed by substrat...More data for this Ligand-Target Pair
TargetCalcium/calmodulin-dependent protein kinase kinase 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataIC50: 6.30nMAssay Description:Inhibition of full-length human GST-tagged CAMKK2 using 5FAM-AKPKGNKDYHLQTCCGSLAYRRR-amide as substrate preincubated for 30 mins followed by substrat...More data for this Ligand-Target Pair
TargetCalcium/calmodulin-dependent protein kinase kinase 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataIC50: 7.90nMAssay Description:Inhibition of full-length human GST-tagged CAMKK2 using 5FAM-AKPKGNKDYHLQTCCGSLAYRRR-amide as substrate preincubated for 30 mins followed by substrat...More data for this Ligand-Target Pair
TargetCalcium/calmodulin-dependent protein kinase kinase 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataIC50: 7.90nMAssay Description:Inhibition of full-length human GST-tagged CAMKK2 using 5FAM-AKPKGNKDYHLQTCCGSLAYRRR-amide as substrate preincubated for 30 mins followed by substrat...More data for this Ligand-Target Pair
TargetCalcium/calmodulin-dependent protein kinase kinase 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataIC50: 7.90nMAssay Description:Inhibition of full-length human GST-tagged CAMKK2 using 5FAM-AKPKGNKDYHLQTCCGSLAYRRR-amide as substrate preincubated for 30 mins followed by substrat...More data for this Ligand-Target Pair
TargetCalcium/calmodulin-dependent protein kinase kinase 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataIC50: 13nMAssay Description:Inhibition of full-length human GST-tagged CAMKK2 using 5FAM-AKPKGNKDYHLQTCCGSLAYRRR-amide as substrate preincubated for 30 mins followed by substrat...More data for this Ligand-Target Pair
TargetCalcium/calmodulin-dependent protein kinase kinase 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataIC50: 13nMAssay Description:Inhibition of full-length human GST-tagged CAMKK2 using 5FAM-AKPKGNKDYHLQTCCGSLAYRRR-amide as substrate preincubated for 30 mins followed by substrat...More data for this Ligand-Target Pair
TargetCalcium/calmodulin-dependent protein kinase kinase 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataIC50: 13nMAssay Description:Inhibition of full-length human GST-tagged CAMKK2 using 5FAM-AKPKGNKDYHLQTCCGSLAYRRR-amide as substrate preincubated for 30 mins followed by substrat...More data for this Ligand-Target Pair
TargetCalcium/calmodulin-dependent protein kinase kinase 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataIC50: 13nMAssay Description:Inhibition of full-length human GST-tagged CAMKK2 using 5FAM-AKPKGNKDYHLQTCCGSLAYRRR-amide as substrate preincubated for 30 mins followed by substrat...More data for this Ligand-Target Pair
TargetCalcium/calmodulin-dependent protein kinase kinase 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataIC50: 13nMAssay Description:Inhibition of full-length human GST-tagged CAMKK2 using 5FAM-AKPKGNKDYHLQTCCGSLAYRRR-amide as substrate preincubated for 30 mins followed by substrat...More data for this Ligand-Target Pair
TargetCalcium/calmodulin-dependent protein kinase kinase 2(Homo sapiens (Human))
GlaxoSmithKline
Curated by ChEMBL
GlaxoSmithKline
Curated by ChEMBL
Affinity DataIC50: 13nMAssay Description:Inhibition of full-length human GST-tagged CAMKK2 using 5FAM-AKPKGNKDYHLQTCCGSLAYRRR-amide as substrate preincubated for 30 mins followed by substrat...More data for this Ligand-Target Pair

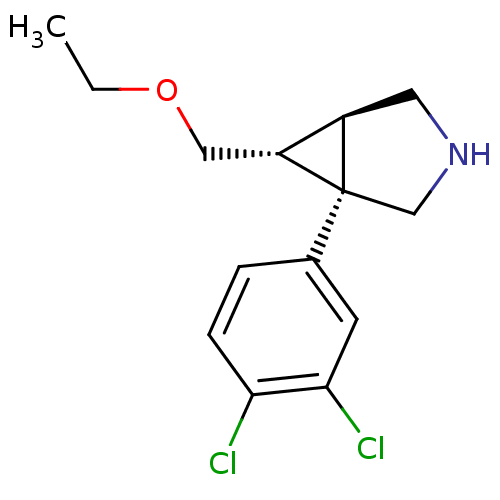
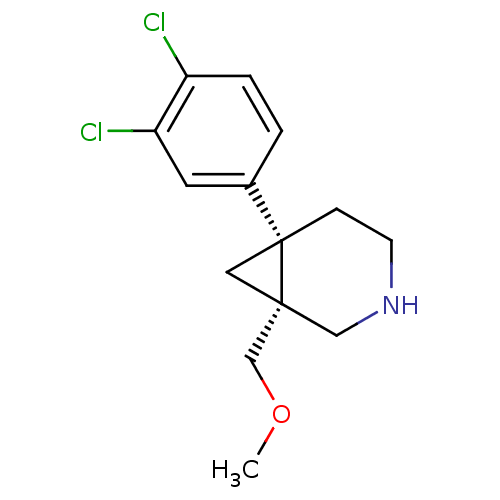
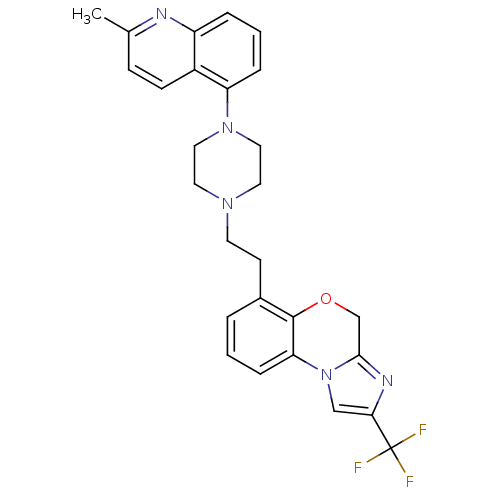
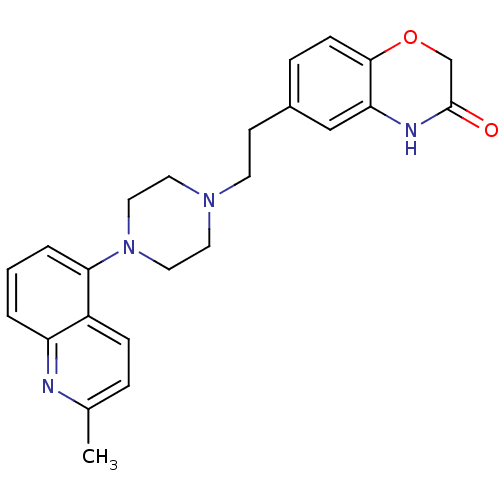


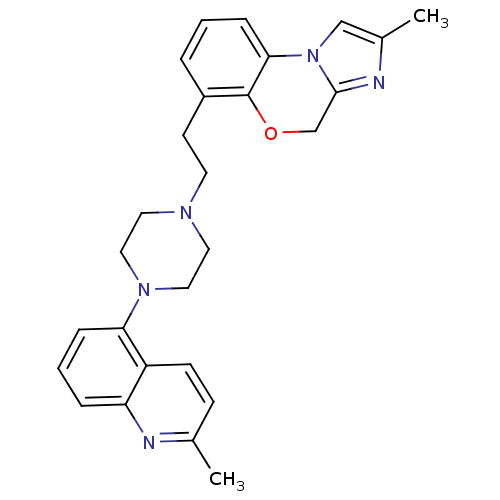

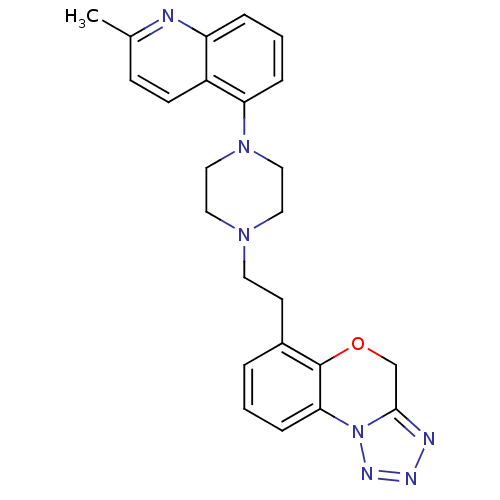
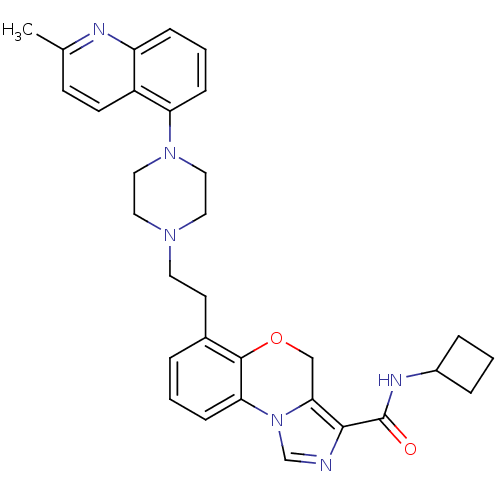


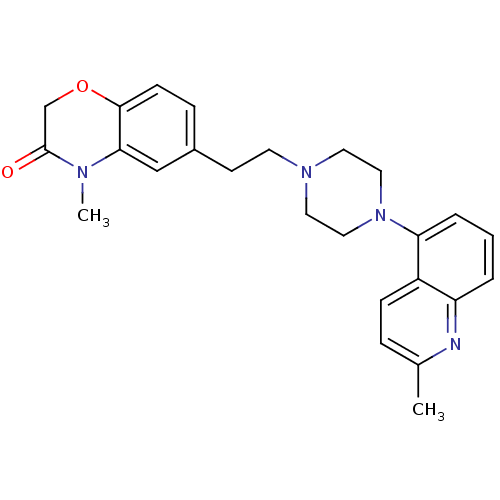

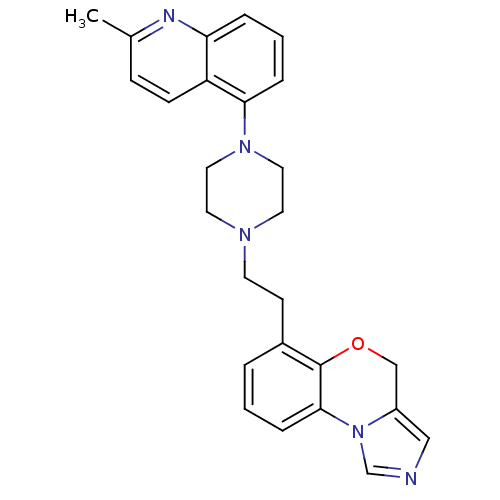
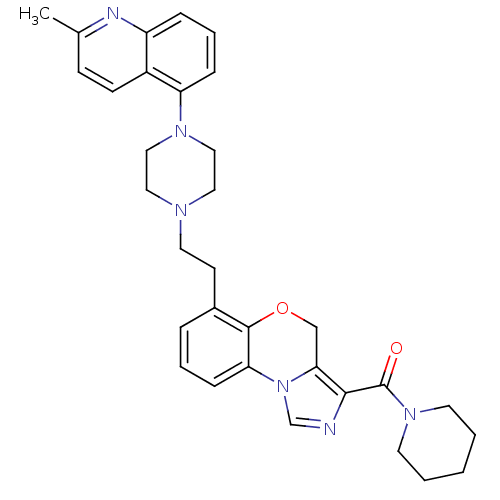
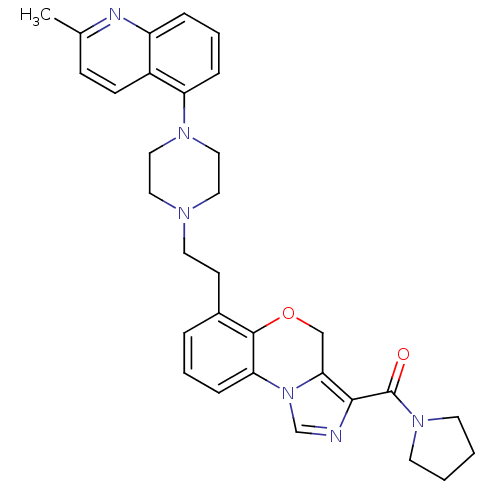




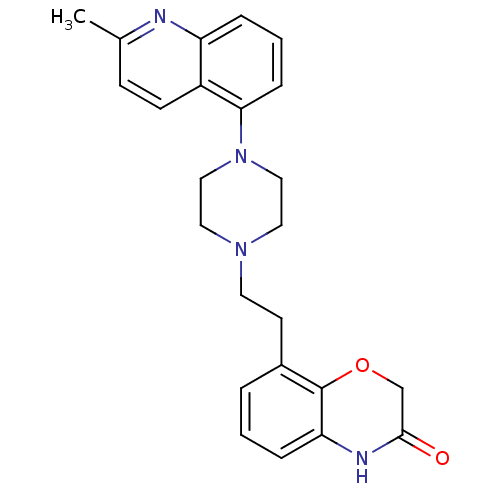
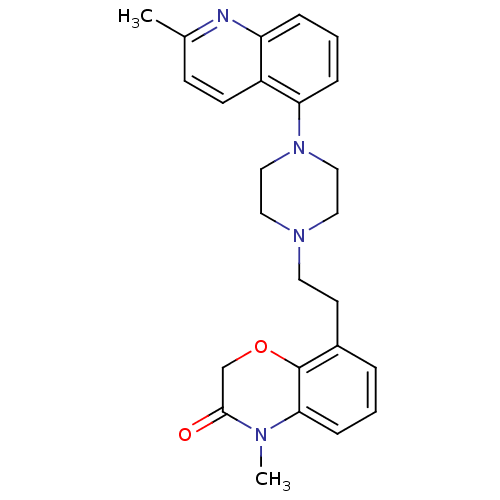
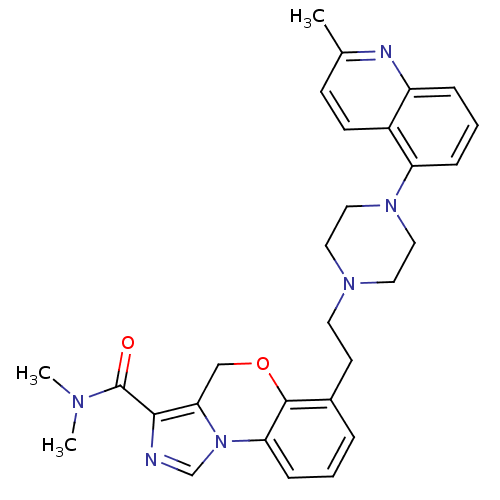






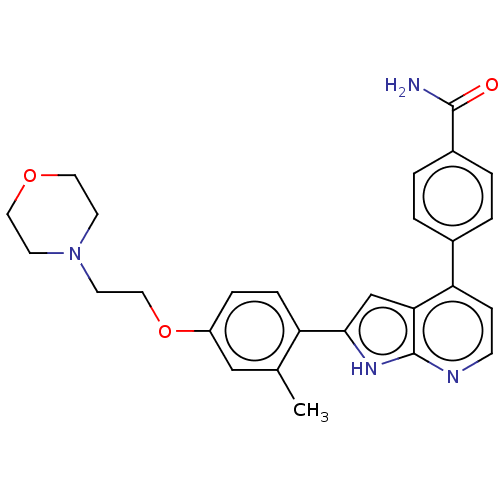

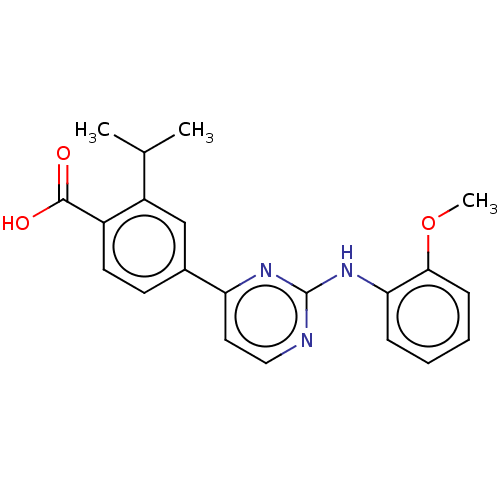
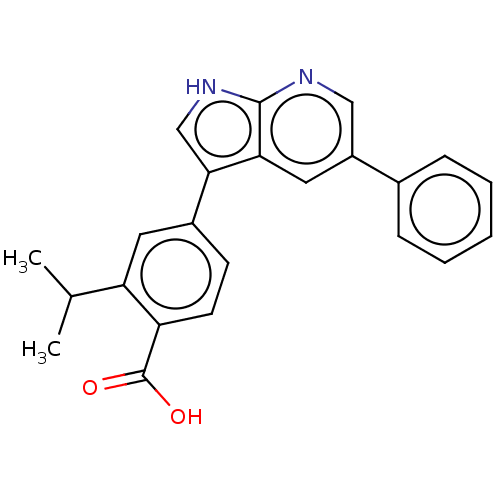

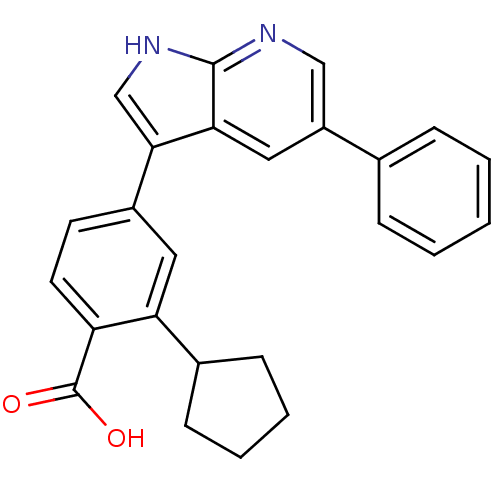
 3D Structure (crystal)
3D Structure (crystal)